Bowel Prep
The Case
The patient is a 73-year-old woman who 20 years ago underwent treatment for breast cancer. At her daughter's suggestion, the patient requested referral for colonoscopy, as she understood there was an increased risk of her having a second malignancy.
At four feet ten inches tall, the patient was petite, weighing only 88 pounds. Her primary care physician referred her to a gastroenterologist whose practice consists of ambulatory endoscopy. In his practice, patients are not routinely seen in advance of their procedures, so the patient called the gastroenterologist's office and was given instructions for her bowel prep. When she inquired about whether the "dose" of the prep needed adjustment given her small size, the nurse told her that this was the "standard" dose. The nurse asked no further questions.
The patient used the bowel prep as instructed, and her husband found her unresponsive on the morning of her scheduled colonoscopy. She was taken to the hospital and found to have significant electrolyte abnormalities including hyponatremia, hypokalemia, hypophosphatemia, hypocalcemia, and hypomagnesemia, all believed to be complications of the bowel prep. She was re-hydrated, her electrolytes were corrected, and she was discharged home after 48 hours.
The Commentary
The patient appears to have had an adverse outcome associated with a bowel preparation for colonoscopy. Given the increasingly frequent use of colonoscopy for screening, it is useful to review the procedures for safely cleansing the bowel. In doing so, I will consider whether this patient's adverse reaction did, in fact, represent an error in the administration of her bowel preparation. I will also discuss the risks of colonoscopy in general, and some procedures for mitigating these risks.
Colon cancer is the third leading cause of cancer death for women in the United States (1). Although this patient's previous history of breast cancer does not increase her risk of colorectal cancer (2), her referral for colonoscopy was appropriate (and, in fact, well overdue), since screening for average-risk adults (male and female) should begin at age 50.
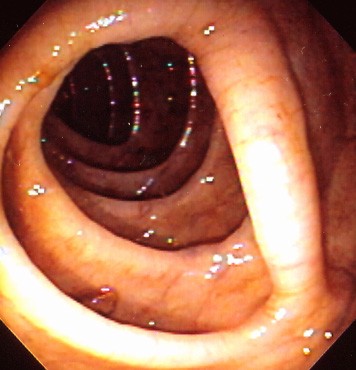
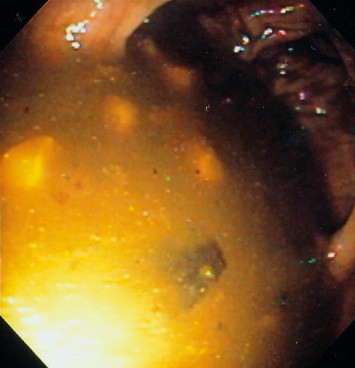
In order to perform safe and accurate colonoscopy, the contents of the colon need to be evacuated, typically referred to as the "bowel prep" (Figure 1). Polyethylene glycol electrolyte lavage solution (PEG-ELS) and oral sodium phosphate (NaP) are two major classes of colonoscopy preparations; each works by a different mechanism. PEG-ELS preparations (such as GoLYTELY) are osmotically balanced, non-absorbable electrolyte solutions that cleanse the bowel by simple washout of ingested fluid without significant fluid and electrolyte shifts. Using this preparation requires that one gallon of the liquid be ingested over a 2-hour period, which some patients may have difficulty completing (both because of the volume and the taste). If the patient is unable to complete the preparation, the bowel will be inadequately prepped (Figure 2), but complications are rare. The major alternatives are hyperosmotic preparations containing sodium phosphate, which draw plasma water into the bowel lumen to achieve the fluid washout. Therefore, the patient must drink other liquids to replace these fluid shifts. Oral sodium phosphate preparations should not be used in patients with renal disease, congestive heart failure, or ascites.(3) A meta-analysis of eight randomized trials comprising 1286 subjects suggested that PEG-ELS and NaP are largely equivalent (including adverse reactions), although the latter may be better tolerated by patients.(4)
Although the specific bowel cleansing regimen given to the patient in this case is not stated, the subsequent electrolyte disturbances reported (hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia) have been associated with oral sodium phosphate-based preparations.(5-8) While the package insert for this over-the-counter (OTC) medication does give a dosage range, it is not weight-based. The only reference to a lower dosage adjustment is for elderly patients, although clinically it is rarely used. In fact, no dosage adjustment was made for age in any of the randomized trials mentioned above, and patient age ranged as high as 90. In practice, the standard dose—45 ml (1.5 oz) given on two occasions prior to the procedure—is used routinely in nearly all patients.
Accordingly, unless this patient had some reason that she was unable to adequately drink fluids, it would be hard to state that an error was made here, except in retrospect. The failure of the nurse to inquire about other medical conditions that might contraindicate this bowel preparation is a potential source of error, although we are not told that the patient had any of these conditions.
Approximately 4-5 million colonoscopic procedures are performed annually in the United States. The benefits of screening in the prevention and detection of colorectal cancer are enormous, allowing for earlier detection of existing cancers, prevention of new cancers, and a reduction in mortality from colorectal cancer. It is estimated that colonoscopic removal of polyps can prevent up to 90% of colorectal cancers.(9) Although the procedure is relatively safe, there are risks associated with the procedure that should be discussed with patients as part of informed consent process. The major complications of colonoscopy are bleeding and perforation; for diagnostic colonoscopy, the overall rate of complications is approximately 0.1%.(10) Although more difficult to place an exact estimate on the rate of complications from the bowel prep itself, the fact that these are largely limited to case reports over the last 30 years, despite the tremendous volume of procedures performed annually, suggests that this is an extremely rare complication.
One wonders whether a pre-procedure visit to the gastroenterologist might have prevented this patient's complication. Most patients are referred for colonoscopy by their primary care physician, who should assess whether the patient's overall medical condition would allow her to benefit from colorectal cancer screening. Prior to the colonoscopy, the endoscopist reviews the patient's medical record to assess the patient's risk. Most institutions send written instructions to the patient regarding the bowel preparation (rather than a pre-procedure visit). If sodium phosphate is used, it is not uncommon for patients to be instructed simply to purchase the OTC medication from their local pharmacy and follow the instructions; PEG-ELS still requires a prescription. Because our hospital has many elderly patients with comorbid conditions that might preclude the use of oral sodium phosphate preparations, we have elected to use a PEG-ELS based regimen for all our patients. Although patients may fail to complete the prep, adverse effects from PEG-ELS are virtually unheard of. If a sodium phosphate prep is used, it may be beneficial to have some sort of pre-procedural encounter to determine if the patient has one of the medical conditions that would preclude the use of this agent, such as renal insufficiency or heart failure.
In light of the present case, it might be tempting to recommend routine pre-procedural (and pre-prep) visits with the gastroenterologist. However, I doubt that such visits would be a judicious use of resources. The United States is presently experiencing a relative shortage of trained gastroenterologists, one that was not predicted as recently as a decade ago. In fact, in 1996 the Gastroenterology Leadership Council, after performing a workforce modeling study that anticipated an oversupply of gastroenterologists, recommended that the number of training positions be decreased by 25%-50%. Unfortunately, that decreased supply has bumped up against an increased demand in recent years. Both patients and the media have become increasingly aware of the benefits of colorectal cancer screening.(11) In light of these benefits, in July 2001, Medicare began to reimburse for screening colonoscopy in average risk individuals, and some third-party payers have followed suit. Pre-procedural physician visits would divert physician resources/productivity from a screening activity with a relatively high yield and proven benefit (colorectal cancer detection and prevention) to an activity with an extremely low yield of questionable benefit, given the overall favorable safety profile of colonoscopy, generally, and bowel preps, specifically.
In summary, this patient appears to have had an adverse event from her bowel preparation, but I don't believe it was related to an error. Preventing similar adverse events in the future may be best accomplished by appreciating the relative contraindications of sodium phosphate preparations; institutions with a high prevalence of cardiac and renal comorbidities (like ours) may find it advantageous to routinely use the alternative PEG-ELS preparation.
Take-Home Points
- More patients are undergoing colonoscopy for colorectal cancer screening.
- Bowel preparation is crucial for effective colonoscopy.
- Two bowel preparations are available that are largely equivalent: Polyethylene glycol electrolyte lavage solution (PEG-ELS) and oral sodium phosphate (NaP). PEG-ELS is remarkably safe, though sometimes poorly tolerated. Sodium phosphate is easier to tolerate, but can cause electrolyte shifts.
- Some have argued for routine pre-procedure visits with gastroenterologists to review the bowel preparation instructions. However, it is unlikely that such visits would be the best use of scarce resources given the excellent safety profile of these regimens.
Douglas B. Nelson, MD Staff Physician in Gastroenterology, Minneapolis VA Medical Center Associate Professor of Medicine, University of Minnesota Medical School
References
1. Jemal A, Murray T, Samuels A, Ghafoor A, Ward E, Thun MJ. Cancer statistics, 2003. CA Cancer J Clin. 2003;53:5-26.[ go to PubMed ]
2. Newschaffer CJ, Topham A, Herzberg, Weiner S, Weinberg DS. Risk of colorectal cancer after breast cancer. Lancet. 2001;357:837-40.[ go to PubMed ]
3. Nelson DB, Barkun AN, Block KP, et al. Technology status evaluation report. Colonoscopy preparations. May 2001. Gastrointest Endosc. 2001;54:829-32.[ go to PubMed ]
4. Hsu CW, Imperiale TF. Meta-analysis and cost comparison of polyethylene glycol lavage versus sodium phosphate for colonoscopy preparation. Gastrointest Endosc. 1998;48:276-82.[ go to PubMed ]
5. Fass R, Do S, Hixson LJ. Fatal hyperphosphatemia following Fleet Phospho-Soda in a patient with colonic ileus. Am J Gastroenterol. 1993;88:929-32.[ go to PubMed ]
6. Vukasin P, Weston LA, Beart RW. Oral Fleet Phospho-Soda laxative-induced hyperphosphatemia and hypocalcemic tetany in an adult: report of a case. Dis Colon Rectum. 1997;40:497-9.[ go to PubMed ]
7. Boivin M, Kahn SR. Symptomatic hypocalcemia from oral sodium phosphate: a report of two cases. Am J Gastroenterol. 1998;93:2577-9.[ go to PubMed ]
8. Beloosesky Y, Grinblat J, Weiss A, Grosman B, Gafter U, Chagnac A. Electrolyte disorders following oral sodium phosphate administration for bowel cleansing in elderly patients. Arch Intern Med. 2003;163:803-8.[ go to PubMed ]
9. Winawer SJ, Zauber AG, Ho MN, et al. Prevention of colorectal cancer by colonoscopic polypectomy. The national polyp study workgroup. N Engl J Med. 1993;329:1977-81.[ go to PubMed ]
10. Nelson DB, McQuaid KR, Bond JH, Lieberman DA, Weiss DG, Johnston TK. Procedural success and complications of large-scale screening colonoscopy. Gastrointest Endosc. 2002;55:307-14.[ go to PubMed ]
11. Cram P, Fendrick AM, Inadomi J, Cowen ME, Carpenter D, Vijan S. The impact of a celebrity promotional campaign on the use of colon cancer screening: the Katie Couric effect. Arch Intern Med. 2003;163:1601-5.[ go to PubMed ]
Figures
Figure 1. Endoscopic View During Colonoscopy With an Excellent Bowel Preparation. Mucosal Details are Easily Seen.
Figure 2. View of a Poor Bowel Preparation. Such Poor Preparation Would Severely Limit The Ability to Detect Polyps and Other Important Pathology During Colonoscopy.