Dangerous Dialysis
Case Objectives
- List common errors that occur in dialysis units.
- Describe steps that can be taken by dialysis units to prevent these common errors.
- Describe the role of the dialysis unit medical director in promoting patient safety in dialysis units.
- List the regulatory agencies with a role in dialysis unit patient safety oversight.
The Case
A 48-year-old man with a long history of diabetes and end-stage renal disease (ESRD) on hemodialysis arrived at his outpatient dialysis center for his scheduled Friday morning session. Before starting dialysis, his nephrologist sat down next to him and stated that a serious error had occurred at the dialysis center. The nephrologist told the patient that, for a number of dialysis sessions, he had been dialyzed using a dialysis membrane that had been inappropriately reused, which meant that he had been exposed to another patient's blood many times.
The dialysis center was actually not sure which dialysis membrane had been reused, so they couldn't identify the specific patients affected by this error. Thus, they were informing all patients who had potentially been exposed to a communicable disease. At this dialysis center, many patients had HIV and hepatitis C, so it was conceivable that this particular patient had been exposed.
The patient was tested for HIV and hepatitis viruses and was treated for 3 months with postexposure prophylaxis for HIV. Ultimately, repeated blood tests were negative for HIV and hepatitis, meaning that the patient did not experience any long-term consequences.
The Commentary
This case illustrates a patient identification error involving dialyzer reuse in a hemodialysis unit. In 2007, more than 500,000 end-stage renal disease (ESRD) patients were dialyzed in the United States, with nearly 65% receiving dialysis in dedicated centers.(1) Most patients undergo hemodialysis three times per week and, in addition to the dialysis treatment, are given multiple medications at the dialysis center (e.g., heparin, erythropoietin stimulating agents, intravenous iron and 1,25 vitamin D analogs, and occasionally antibiotics). Thus, dialysis centers can be uniquely hazardous places for patients. Common errors in dialysis units (Table 1) may involve the dialysis procedure, including errors in patient identification, the dialysis equipment (dialyzer, dialysis solution), the patient's dialysis access (arteriovenous fistula, graft, or indwelling venous catheters), or medications administered during the treatment.(2,3) In addition, in 2007, 81,000 dialysis patients were older than 75 years.(1) Many elderly patients have impaired ambulation and/or poor functional status, and thus there is a higher risk of falls, especially in the setting of hemodialysis-associated hemodynamic changes.(4) The recognition and reduction of errors in dialysis units are mandated by oversight bodies certifying dialysis units (Table 2) and increasingly are among the responsibilities of dialysis unit medical directors (Table 3).(5,6)
Incorrect Dialyzer or Dialysis Solution and Water Quality Issues
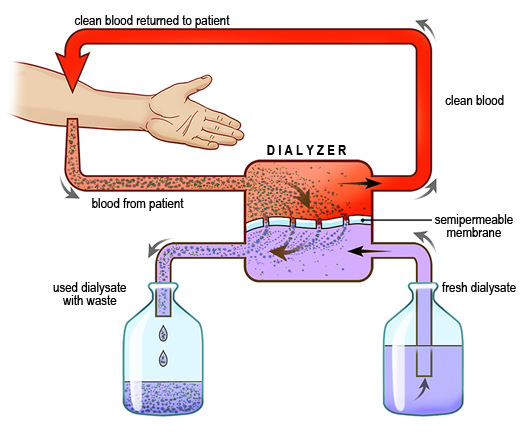
Hemodialysis removes small solutes (potassium, blood urea nitrogen [BUN], creatinine, etc.) from patients, using a system in which the patient's blood is pumped through a semipermeable membrane (the dialyzer) and the dialysis solution (dialysate) flows countercurrent to the blood, resulting in the movement (diffusion) of solutes (Figure). Potential sources for errors exist throughout this system, including contamination of the dialysate and/or the water used to make the dialysate, use of an incorrect dialysate for a given patient, ill-fitting lines connecting the dialyzer, dialyzer leaks, and issues with the patient's access (arteriovenous fistula, graft, or venous catheter). Hemolysis, transmission of infection, and accumulation of toxins have all been reported.(3) In some cases, incorrect dialysis solution or contaminated water was the source of the adverse event; in others, human error contributed.
In the 1980s, the proportion of dialysis centers reusing dialyzer membranes increased, peaking in 1997 when 80% of centers reused dialyzers.(7) Such reuse involves sterilizing the dialyzer after each dialysis treatment and then reusing it for subsequent treatments in the same patient. Reusing dialyzers lowers the cost of a dialysis treatment and reduces the amount of biomedical waste generated. However, reusing dialyzers, which is safe if done correctly, also creates opportunities for medical errors—as demonstrated by this case. A formal system to identify each patient and match the specific membrane to the individual patient is required to avoid exposing patients to dialyzers used by other patients. Standards for dialyzer reuse programs were developed by the Association for the Advancement of Medical Instrumentation (AMMI), the same body setting the standards for dialysis unit water.(8) Reuse standards include protocols to reduce the risk of errors, including standards addressing patient identification, dialyzer labeling, and environmental and processing procedures.
As this case illustrates, human error, combined with a weak system, is often at the root of adverse events. When multiple patients are completing dialysis treatments at the same time and a new shift of patients is beginning treatment shortly thereafter, opportunities for dialyzer mislabeling or failing to match a dialyzer with the appropriate patient exist. The patient and dialysis treatment volumes vary significantly and depend on the individual hemodialysis unit census. Large units may have as many as 30 patients or more dialyzing per shift with 3 to 4 shifts per day, resulting in as many as 90 or more treatments per day, or more than 500 treatments per week. Barcode labeling of dialyzers; staff education; avoiding reuse in patients with infectious diseases; specific protocols for the collection, storage, and cleaning of used dialyzers; and staggering dialysis treatments to limit the number of patients initiating therapy at the same time may all reduce the potential for errors.(9) At the peak of reprocessing programs for dialyzers, up to 26% of patients refused to participate in reuse programs (reuse can only be done with informed consent of the patient).(10) Recently, with the development of more biocompatible dialyzers and many dialysis providers favoring single-use dialyzers, the proportion of dialysis units employing a reuse program has fallen. In 2005, only 40% of dialysis units practiced dialyzer reuse.(9) Thus, the primary reason for maintaining a reuse program today is economic, and some would argue that the best practice is to abandon reuse entirely.(9) Considering the risks for errors associated with dialyzer reuse programs and the falling economic incentives to practice reuse, ongoing decline of reuse programs is to be expected.
Patient Falls and Medication Errors
In addition to errors with the dialyzer or dialysis solution, a Renal Physicians Association survey revealed that medication errors and patient falls can also be safety issues at dialysis centers.(11) In this survey, 87% of dialysis unit staff acknowledged a mistake or error (of any kind) in a dialysis treatment in the last 3 months. Although there is no comprehensive published study on the prevalence of errors in dialysis centers, one retrospective study identified 88 errors in 64,541 treatments in 4 dialysis units, resulting in 1 error or adverse event every 733 treatments.(2) A medication error occurred every 2,151 treatments, and most (24 of 35 or 69%) were errors of omission (i.e., the prescribed medication was not given).(2)
Patient falls are a fairly common adverse event in dialysis units.(2,4) In one study, 47% of dialysis patients fell over 190.5 person-years of follow-up.(4) Risk factors for falls included patient age, number of comorbidities, having a lower systolic blood pressure before starting dialysis, and a history of falls.(4) In addition, after dialysis sessions, hemodynamic shifts can lead to transient hypotension (low blood pressure) and dizziness.(4) The fall risk in hemodialysis patients is higher than in the general population.(4) Assessment of fall risk, exercise programs to increase muscle strength, reducing the use of neuropsychiatric medications, and avoiding hypotension in dialysis patients may all reduce fall risk.(4,11)
Oversight and the Medical Director's Role in Dialysis Unit Patient Safety
Dialysis units operate under multiple regulatory bodies overseeing patient safety as part of their responsibilities (Table 2). Under the Centers for Medicare & Medicaid Services (CMS) 2008 revisions, conditions for Medicare coverage for dialysis specify that each dialysis facility measure, analyze, and track quality indicators reflecting facility operations and processes of care.(5) Dialysis facilities must comply with these conditions to receive Medicare payments. The quality indicators include identification of medical injuries and medical errors. Increasingly, the dialysis unit medical director's responsibilities include leading the center's quality assurance program.(5,6) The 2008 Medicare revisions effectively shifted the medical director's responsibilities from implementation of policies and procedures to assurance that the staff (including attending physicians) adhere to mandated policies and procedures and that the unit's policies and practices result in favorable patient outcomes.(5) The CMS conditions of coverage incorporated the AAMI standards for dialysate and water, guidance on the reuse of dialyzers, and the Centers for Disease Control and Prevention's standards for infection control and safe use of intravascular catheters.(5) The medical director is responsible for ensuring that facility policies, procedures, monitoring, and auditing are congruent with these standards and that the dialysis unit staff is adequately trained in the policies and procedures (Table 3). Surveyors from state departments of health can close dialysis units that are not in compliance with standards of care.
Facility oversight is also provided directly through the ESRD Networks. Members of the 18 current geographically based networks participate in facility quality improvement projects under the supervision of CMS. Recently, two regional networks focused specifically on patient safety, and their work may provide guidance for future dialysis facility quality improvement projects.(12,13)
Take-Home Points
- The potential for adverse events and medical errors in hemodialysis units is high due to the procedure itself, the need for medications, the risk for falls, and the comorbidities of the patient population.
- Safety in the hemodialysis unit is under the oversight of the facility's medical director and manager, supervised by the ESRD Networks, state, and national organizations under CMS.
- Reducing errors in dialysis units is a focus of quality improvement projects supported by the ESRD Networks and is mandated by CMS under Medicare's conditions for coverage.(9)
- Future investigation into the occurrence and prevention of adverse events and errors in dialysis units is needed.
Jean L. Holley, MD Clinical Professor of Medicine
University of Illinois, Urbana-Champaign
Carle Physicians Group
Faculty Disclosure: Dr. Holley has declared that neither she, nor any immediate member of her family, has a financial arrangement or other relationship with the manufacturers of any commercial products discussed in this continuing medical education activity. In addition, the commentary does not include information regarding investigational or off-label use of pharmaceutical products or medical devices.
References
1. US Renal Data System, USRDS 2009 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD; 2009. [Available at]
2. Holley JL. A descriptive report of errors and adverse events in chronic hemodialysis units. Nephrol News Issues. 2006;29:57-58, 60-61, 63. [go to PubMed]
3. Kliger AS. Patient safety in the dialysis facility. Blood Purif. 2006;24:19-21. [go to PubMed]
4. Cook WL, Tomlinson G, Donaldson M, et al. Falls and fall-related injuries in older dialysis patients. Clin J Am Soc Nephrol. 2006;1:1197-1204. [go to PubMed]
5. DeOreo PB. The medical directorship of renal dialysis facilities under the new Medicare conditions for coverage: challenges and opportunities. Blood Purif. 2009;27:16-21. [go to PubMed]
6. Maddux FW, Maddux DW, Hakim RM. The role of the medical director: changing with the times. Semin Dial. 2008;21:54-57. [go to PubMed]
7. Finelli L, Miller JT, Tokars JI, Alter MJ, Arduino MJ. National surveillance of dialysis-associated diseases in the United States, 2002. Semin Dial. 2005;18:52-61. [go to PubMed]
8. AAMI Standards. Arlington, VA: Association for the Advancement of Medical Instrumentation. [Available at]
9. Lacson E Jr., Lazarus JM. Dialyzer best practice: single use or reuse? Semin Dial. 2006;19:120-128. [go to PubMed]
10. Okechukwu CN, Orzol SM, Held PJ, et al. Characteristics and treatment of patients not reusing dialyzers in reuse units. Am J Kidney Dis. 2000;36:991-999. [go to PubMed]
11. Report on Findings From the ESRD Professional Survey, March 2007. Rockville, MD: Renal Physicians Association; 2007. [Available at]
12. 5-Diamond Patient Safety Program. Midlothian, VA: Mid-Atlantic Renal Coalition. [Available at]
13. 5-Diamond Patient Safety Program. Woodbridge, CT: Network of New England. [Available at]
Tables
Table 1. Common Errors Occurring in Dialysis Units.
|
Incorrect dialyzer/dialysis solution/water quality issues |
|
|
|
| Patient falls |
| Medication errors and omission |
| Nonadherence to procedures |
| Vascular access-related events |
|
|
|
Table 2. Organizations Involved in Oversight of Patient Safety in Hemodialysis Units.
| Centers for Medicare & Medicaid Services (CMS) | Conditions of coverage include specific quality indicators. |
|---|---|
| Surveyors from State Department of Health | Can close units that do not meet standards of care. |
| End-Stage Renal Disease (ESRD) Networks | 18 geographically organized committees under CMS supervision. Responsible for quality assurance and improvement in dialysis facilities within the network. Medical directors are given periodic assessments of unit morbidity and mortality compiled by the networks through CMS. |
| United States Renal Data System (USRDS) | A repository of data collected on each chronic dialysis patient covered under Medicare. An annual yearly report is published including special studies examining morbidity and mortality of dialysis. Data provided by USRDS may determine ESRD Network quality improvement projects and lead to development of standards of care. |
| Association for the Advancement of Medical Instrumentation (AAMI) | Sets standards for water quality, dialysate, and reuse procedures. |
| Centers for Disease Control and Prevention (CDC) | Sets standards for infection control in dialysis units and provides standards for indwelling catheter-associated infections. |
Table 3. Additional Factors in Oversight of Safety within Dialysis Facility.
| Dialysis Facility |
| Dialysis Facility Governing Body |
| Dialysis Unit Medical Director |
|
|
|
|
| Dialysis Unit Manager |
| Patients and dialysis unit staff |
Figure
Figure. The hemodialysis process.(Illustration © 2010 by Chris Gralapp.)