Failure to Report
Case Objectives
- List common causes of medical errors.
- Appreciate the magnitude of underreporting of adverse events.
- List the common barriers to reporting adverse events and near misses.
- Discuss steps that individuals and institutions can take to increase reporting.
Case & Commentary: Part 1
A well-appearing 9-month-old infant weighing 8 kg presented with urinary frequency and white cells in her urine. The emergency department (ED) physician ordered Rocephin (the brand name for the antibiotic ceftriaxone), 450 mg intramuscularly (IM), for empiric treatment of a urinary tract infection (UTI) to be given immediately.
Several hours later, when removing another vial of ceftriaxone from the automated dispensing cabinet (Pyxis), the nurse noted that there were two vials in the drawer instead of the expected one. In the medicine administration area, the nurse found a partially empty vial of parenteral cefazolin, a different antibiotic that had not been ordered for any patient that night. On the top of the vial was a chalky dried substance, and the admixture remaining in the bottle was cloudy. In this institution, ceftriaxone is routinely mixed with 1% lidocaine to decrease postinjection pain. The resulting mixture is clear and colorless. It was concluded that the infant most likely received 450 mg of IM cefazolin instead of the intended 450 mg of ceftriaxone.
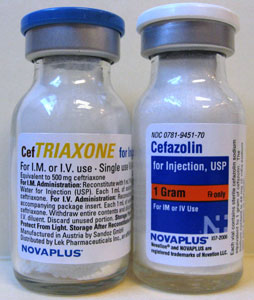
The medications are routinely ordered by the ED physicians as "Rocephin" and "Kefzol" but stocked as "ceftriaxone" and "cefazolin" (Figure). The vials, in the three sizes used to make the admixtures, are all contained in the same Pyxis drawer.
The error-producing conditions that most likely contributed to this mistake—similarly named medications stored in close proximity and an environment predisposed to distractions—should be recognizable to caregivers, as these factors are common causes of medication administration mistakes.(1) These conditions increased the likelihood that the nurse would get the wrong medication from the automated dispenser. Retrieving medications is something nurses do many times during a shift, and, most of the time, it goes as planned. In the few instances when the wrong medication is obtained, it is most often recognized and corrected before administration occurs. In this case, the usual safeguards such as double-checks and read-backs failed, allowing the error to reach the patient. What was different this time? An interview with the nurse involved might provide a clear answer, although the mistake would most likely be attributed to an unexplainable lapse in following procedures.
The nurse's failure to confirm the medication's identity prior to administration falls into the category of an "unsafe act." Merely telling the nurse to be more careful next time does little to improve the safety of the system, especially when the nurse involved in this event is probably the least likely person to repeat the error. To prevent unintentional unsafe acts—slips, lapses, mistakes, or procedure violations—the contributory factors and latent conditions must be resolved.
The contributory factors in this event fall into the environmental category. Some hazards in this category are easier to overcome than others. Periodic pharmacy inspections of all drug storage areas, including automated dispensing cabinets, can be used to identify and resolve unsafe conditions.(2) Separation of inventory will reduce mix-ups caused by look-alike/sound-alike drugs.(3)
The safety hazards caused by a distracting environment are not as easily fixed. Interruptions during drug retrieval may be minimized by creating a zone of safety around the medication storage area. This can be done by either moving medication storage to a quieter location or creating some type of "Do Not Disturb" signage that is posted by the storage area during times when medications are being retrieved. Facilities using this strategy have found that people are less likely to disturb a nurse preparing a medication for a patient when there is a visual reminder.(4,5)
The latent conditions underlying this event must be understood by asking why these situations were allowed to exist. In other words, why are sound-alike drugs stored in close proximity and why haven't steps been taken to reduce cognitive overload on caregivers even though it is well known that both of these factors contribute to medication errors? Often, complacency is the culprit. When the work environment is normally reliable, safe, and accurate, it is easy for people to become complacent—a belief that past experiences will repeat themselves in current situations. Trusting that "the process will not fail" or "a problem will not happen to me" is a latent condition that causes individuals to let down their guard. This trust in the system can also contribute to organizational complacency, creating an environment that lacks a sense of urgency about patient safety.
If nothing is done to reduce complacency, it will undermine the effectiveness of tactics aimed at resolving the contributory factors. Overcoming individual and organizational complacency requires an ongoing dialogue about patient safety with the goal of creating greater awareness of what can go wrong and greater willingness to reduce potential risks and safety hazards.(6)
Case & Commentary: Part 2
The nurse who had given this medication was very upset and spoke with the ED physician on duty about the event, as 450 mg of IM cefazolin is an overdose for an 8-kg baby. The nurse was informed that the medications were essentially equivalent and did not pursue the matter further or report the error to her supervisor or through the institution's incident reporting system.
When the physician failed to acknowledge the potentially harmful medication error, caregivers missed a chance to protect future patients from harm. Undoubtedly, this was not the physician's intention, yet this is a common "knee jerk" reaction following an error. While some observers have attributed this reaction to the medical profession's "conspiracy of silence" (7,8), it is actually a very natural human impulse to hide mistakes so that potential problems or conflicts can be avoided. Couple this natural impulse with professional attitudinal barriers and individual feelings of helplessness, fear, and anxiety, and we have a situation in which it is difficult to admit that a mistake occurred. Without interviewing the physician in this case, the reason for not acknowledging the error is unknown. Because health care professionals often treat their mistakes as personal failures, I'd speculate that the physician may have wanted to protect the nurse and him- or herself from feelings of guilt.(9)
This is an unfortunate case from two perspectives. First, the child received an overdose of antibiotics. By not acknowledging the mistake, the health care team made no attempt to inform the parent of what happened so that potential effects could be mitigated. Second, and just as important, a learning opportunity was lost. How many other patients must be harmed by similar mistakes before the factors that led to the mistakes are fixed?
Learning could have occurred at two levels. Even if a formal incident report was not completed, the physicians and nurses in the unit could have participated in a frank discussion about what had happened and how future mistakes of this type could be averted. One tool drawn from crew teamwork training in aviation is self-critiquing, a team activity that involves debriefing of recently completed activities.(10,11) Such discussions also foster a team awareness of failures, which can help reduce complacency—a probable latent condition in this case. Instead, the message that may have been imparted is, "When things don't go right, hide the mistake" rather than "When things don't go right, openly discuss how to keep it from happening again."(12) By completing an incident report for this event, the learning opportunity would have been expanded to the entire organization.
It is impossible to determine the rate of unacknowledged medical mistakes, especially in the absence of an untoward outcome. Even when caregivers concede that a mistake has been made, it often goes unreported—impeding organizational learning and often individual learning. Underreporting of adverse events is estimated to range from 50% to 96% annually (13,14), and underreporting of no-harm or "near miss" errors is even greater.(15) How to improve reporting has become a much-researched question in recent years, with the reasons for nonreporting found to be many and varied.(16)
How can health care organizations make reporting a regular part of individual practice? It is tempting to persuade compliance with arguments such as:
- "It's your professional duty."
- "The incident database will help identify improvement opportunities."
- "We can see if our safety improvement efforts are making a difference."
While these are convincing reasons to report incidents, they may not be persuasive enough. For an individual to report an incident, the most important factor is safety—not the patient's, but his or her own. The people must feel safe from undeserved disciplinary action or retaliation. A study of hospital nursing units found that higher reporting rates were correlated with unit members' perception of the risk of discussing mistakes openly.(17) Where there was a climate of fear, willingness to report mistakes was reduced. Other factors that are known to influence reporting (18) are:
- uselessness (perceived attitudes that management would take no notice and was not likely to do anything about the problem);
- acceptance of risk (incidents are part of the job and cannot be prevented); and
- practical reasons (too time consuming or difficult to submit a report).
To encourage reporting, organizations need to minimize these factors and create a forgiving environment in which people feel "psychologically safe" to acknowledge errors. Strategies that have proven to be effective are summarized in the Table. These strategies are central to the success of many incident reporting initiatives. For example, in the 2 years following addition of a 24-hour call center, incident reports submitted to the South Australia Department of Health increased by 275%.(19) The rise in reports was attributed to enhanced reporting accessibility as well as the new culture of openness. A nonpunitive culture and assurance of reporter confidentiality are two factors that have influenced the success of the incident reporting system at the Veterans Health Administration. In the first 5 years since its inception, more than 140,000 incident reports were submitted.(20) Availability of a secure Internet site for anonymous event reporting contributed to a high rate of submissions to the neonatal intensive care incident reporting project sponsored by the Vermont Oxford Network.(21) During patient safety executive walkarounds, leaders at Kaiser Permanente San Diego (CA) Medical Center personally speak with physicians and staff about the importance of learning from mistakes. Leaders' ongoing reinforcement of the organization's "just culture" has contributed to an increase in reporting and discussion of errors and near misses.(22) Visible improvements are another factor influencing reporting in the Kaiser Permanente initiative. Physicians and staff see that when incidents are reported, problems do get fixed.(23)
Take-Home Points
- The majority of adverse events go unreported.
- Near misses and adverse events provide valuable teaching opportunities for caregivers and organizations.
- Caregivers should incorporate reporting into routine practice.
- Organizations should work to create a safe environment that encourages reporting and provides a streamlined, user-friendly process.
Patrice L. Spath, BA, RHIT Healthcare Quality Specialist Brown-Spath & Associates Forest Grove, Oregon
Faculty Disclosure: Dr. Spath has declared that neither she, nor any immediate member of her family, has a financial arrangement or other relationship with the manufacturers of any commercial products discussed in this continuing medical education activity. In addition, the commentary does not include information regarding investigational or off-label use of pharmaceutical products or medical devices.
References
1. Committee on Identifying and Preventing Medication Errors, Institute of Medicine; Aspden P, Wolcott JA, Bootman JL, Cronenwett LR, eds. Preventing Medication Errors. Washington, DC: National Academy Press; 2007.
2. Weber RJ, ed. The Handbook on Storing and Securing Medications. Oakbrook Terrace, IL: Joint Commission Resources and the American Society of Health-System Pharmacists; 2006.
3. Medication Errors. HRC Risk Analysis: Pharmacy and Medications 1. Plymouth Meeting, PA: ECRI; November 2004. Available at: www.ecri.org/Include/Docs/Marketing/HRC_MedErrors.pdf.
4. Pape TM. Applying airline safety practices to medication administration. Medsurg Nurs. 2003;12:77-93. [go to PubMed]
5. Hodgkinson B, Koch S, Nay R, Nichols K. Strategies to reduce medication errors with reference to older adults. Int J Evid Based Healthc. 2006;4:2-41.
6. Weick KE, Sutcliffe KM. Managing the Unexpected: Assuring High Performance in an Age of Complexity. Vol 1. New York: John Wiley and Sons; 2001.
7. Lens P, Van De Wal G, eds. Problem Doctors: A Conspiracy of Silence. Amsterdam, NLD: IOS Press; 1997.
8. Leven L, Ashton JR. The conspiracy of silence in health care quality. J Epidemiol Community Health. 2006;60:913.
9. Wu AW. Medical error: the second victim. The doctor who makes the mistake needs help too. BMJ. 2000;320:726-727. [go to PubMed]
10. Wilson KA, Burke CS, Priest HA, Salas E. Promoting health care safety through training high reliability teams. Qual Saf Health Care. 2005;14:303-309. [go to PubMed]
11. Risser DT, Simon R, Rice MM, Salisbury ML. A structured teamwork system to reduce clinical errors. In: Spath PL. Error Reduction in Health Care: A Systems Approach to Improving Patient Safety. San Francisco, CA: Jossey-Bass; 1999:235-278.
12. Singer PA, Wu AW, Fazel S, McMillan J. An ethical dilemma: medical errors and medical culture. BMJ. 2001;322:1236-1240. [go to PubMed]
13. Committee on Quality of Health Care in America, Institute of Medicine; Kohn L, Corrigan J, Donaldson M, eds. To Err is Human: Building a Safer Health System. Washington, DC: National Academy Press; 2000.
14. Joshi MS, Anderson JF, Marwaha S. A systems approach to improving error reporting. J Healthc Inf Manag. 2002;16:40-45. [go to PubMed]
15. Bates DW, Boyle DL, Vander Vliet MB, Schneider J, Leape L. Relationship between medication errors and adverse drug events. J Gen Intern Med. 1995;10:199-205. [go to PubMed]
16. Vincent C, Stanhope N, Crowley-Murphy M. Reasons for not reporting adverse events: an empirical study. J Eval Clin Pract. 1999;5:13-21. [go to PubMed]
17. Edmondson AC. Learning from mistakes is easier said than done: group and organizational influences on the detection and correction of human error. J Appl Behav Sci. 1996;32:5-28.
18. van der Schaaf T, Kanse L. Checking for biases in incident reporting. In: Phimister JR, Bier VM, Kunreuther HC. Accident Precursor Analysis and Management: Reducing Technological Risk Through Diligence. Washington DC: National Academic Press; 2004:119-126.
19. Error Reporting on the Increase—South Australian Department of Health [news release]. Adelaide, SA, Australia: Patient Safety International; May 3, 2006. Available at: http://www.patientsafetyint.com/updates.aspx?ID=7. Accessed March 1, 2007.
20. McCarthy D, Blumenthal D. Committed to Safety: Ten Case Studies on Reducing Harm to Patients. New York, NY: The Commonwealth Fund; April 2006. Publication No. 923. Available at: http://www.cmwf.org/usr_doc/McCarthy_safetycasestudies_923.pdf. Accessed March 1, 2007.
21. Suresh G, Horbar JD, Plsek P, et al. Voluntary anonymous reporting of medical errors for neonatal intensive care. Pediatrics. 2004;113:1609-1618. [go to PubMed]
22. Feitelberg SP. 2005 Lawrence Patient Safety Award Winner: Patient Safety Executive Walkarounds. Permanente J. Summer 2006;10:29-35.
23. Spath PL. Sustainable patient safety. H&HN Online. December 12, 2006. Available at: http://www.hhnmag.com/hhnmag_app/jsp/articledisplay.jsp?dcrpath=HHNMAG/PubsNewsArticle/data/2006December/ 061212HHN_Online_Spath&domain=HHNMAG. Accessed March 1, 2007.
Table
Strategies to Improve Incident Reporting
| Promote and sustain a culture of learning from mistakes. |
| Make incident reporting an individual performance expectation. |
| Have clear definitions for reportable events that everyone understands. |
| Make it easy to report and provide several different reporting methods. |
| Allow for anonymous error reporting. |
| Maintain a confidential incident reporting system. |
| Use reports to identify common error-producing factors, not just to create incident counts. |
| Share the learning derived from incident analyses with physicians and staff. |
| Communicate and celebrate improvements that result from analyzing reported events. |
Figure
Figure. Example of Similar-Looking Vials.