Safeguarding Diagnostic Testing at the Point of Care
Kost GJ, Ehrmeyer SS. Safeguarding Diagnostic Testing at the Point of Care. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2017.
Kost GJ, Ehrmeyer SS. Safeguarding Diagnostic Testing at the Point of Care. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2017.
The Case
A 23-year-old woman presented to the family medicine clinic for contraception. She was sexually active with one partner and expressed interest in long-term contraception. After talking with her provider, she selected intrauterine device (IUD) placement.
The protocol in the clinic was to check a urine pregnancy test prior to placing an IUD. The patient provided a urine sample to the nurse. Point-of-care testing in the office revealed a negative result. The physician informed the patient and then placed the IUD during the same visit without complication.
At the end of her shift, the nurse manually entered the results of all point-of-care tests performed in the office that day into the appropriate patient's electronic medical record. By mistake, she entered the result of the urine pregnancy test for this patient as positive instead of negative. When the patient's provider received the positive result via electronic notification, she recognized the error and immediately corrected the medical record.
Although the mistake was recognized quickly and no incorrect information was relayed to the patient, the provider decided to review the results of all point-of-care tests that had been manually entered into the electronic health record system for her patients that day to make sure there were no other errors.
The Commentary
by Gerald J. Kost, MD, PhD, MS, and Sharon Ehrmeyer, PhD
This case highlights important practice principles for point-of-care testing, defined as medical testing at or near the site of patient care.(1) Although we are not certain how many errors occur when tests are performed at the point of care, it is clear that the volume of point-of-care tests is increasing rapidly and will continue to do so worldwide.(2) In accordance with the United States Clinical Laboratory Improvement Amendments (CLIA) of 1988, the CLIA Program establishes lab testing standards and certifies clinical labs. For certain types of tests deemed to be lower risk, a certification waiver can be obtained. The simplest point-of-care tests, such as the urine pregnancy test described in this case, can be performed under such a certificate of waiver.
As of June 2016, certificate of waiver test sites constitute 70% of the 254,000 CLIA-registered testing sites in the US, according to the Centers for Medicare and Medicaid Services (CMS). In outpatient primary care, the most frequently performed point-of-care tests probably are urinalysis with dipstick followed by urine culture only if the urinalysis is positive, and rapid strep followed by a throat culture if the rapid test is negative. A huge variety of tests are available, and some perform more reliably than others. It is up to the health care organization to first evaluate and then select the appropriate methodology or device for the testing situation. Under a CLIA waiver, the provider or organization must follow the current manufacturer's instructions for waived testing, but the test is not subject to any additional standards or regulations. Organizations typically mandate safety precautions (e.g., Occupational Safety and Health Administration [OSHA]). Routine safety precautions include safe handling of patient specimens, safe disposal of test reagents after use, and wearing proper attire such as gloves.
In 2005, the Centers for Disease Control and Prevention (CDC) identified quality and safety concerns for waived tests.(3) The top three errors cited as impacting patient health included: (i) lack of current and updated manufacturers' instructions, (ii) failure to follow manufacturers' instructions, and (iii) reporting of incorrect results. Using improperly stored or outdated reagents, mixing reagents from different kits or lots, using incorrect value assignment from package inserts, not documenting relevant information including results, and inadequate training and lack of knowledge about good laboratory practices were other factors included on the CDC list.
Waived testing consists of two categories: kits and instrumentation. Kits require manual result entry into middleware or the patient record, as seen with the urine pregnancy test described above. Using manual data entry for patient results inevitably generates mistakes. Most instrumented tests possess the capability to automatically download test results directly (or through middleware) into the patient record. Systematic process checks and the capability to directly download test results help mitigate transcription errors. Fortunately, in this case the error was caught promptly, to the credit of the nurse and provider.
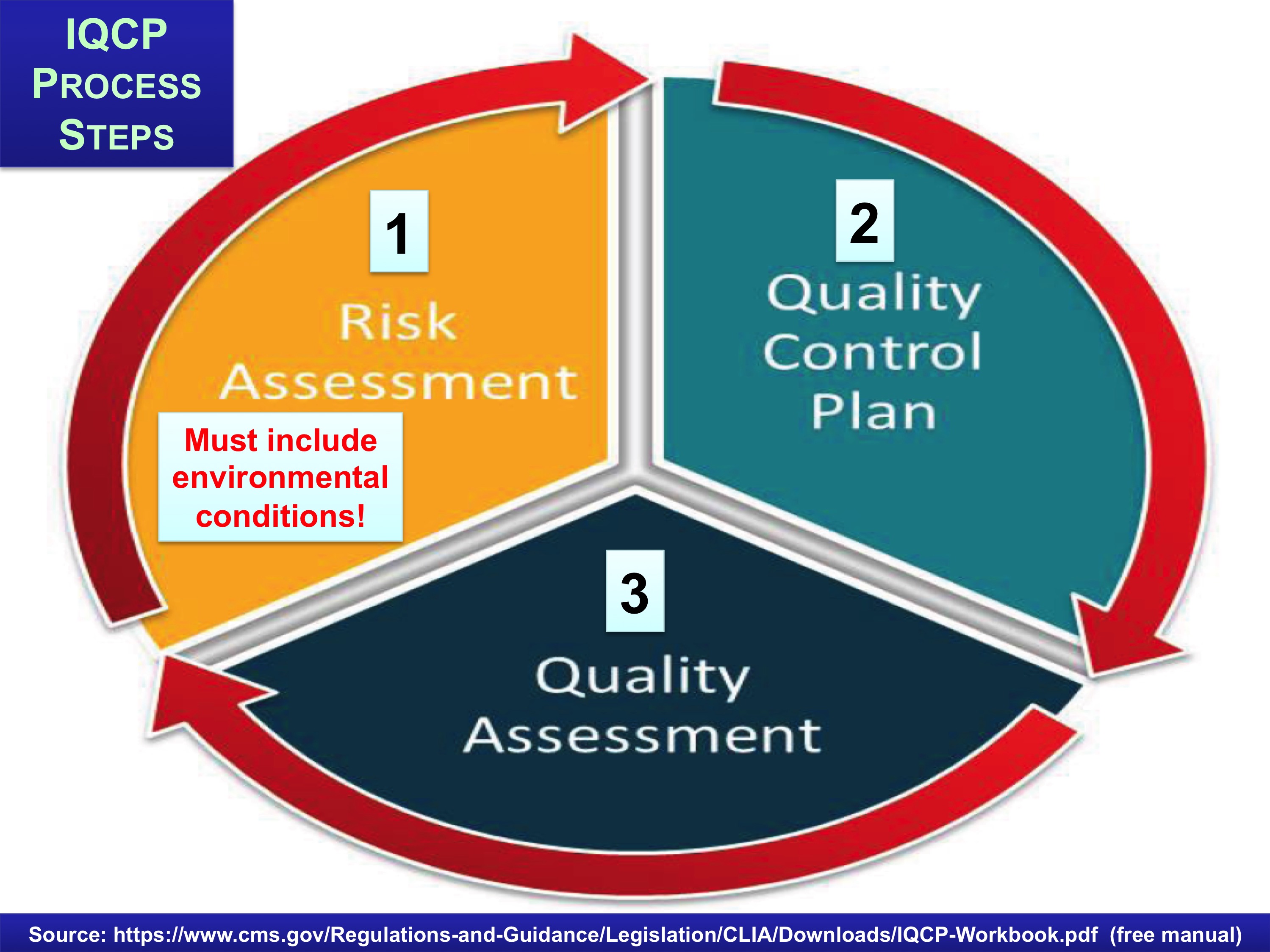
However, it is hazardous to depend on the vigilance of busy providers to prevent errors. In this clinic, it would be advisable for the provider and nurse to develop an individualized quality control program (IQCP) for checking results (4) to ensure ongoing quality. An IQCP includes three phases spanning the entirety of the testing process: risk assessment, quality control plan, and quality assessment (Figure 1).(5-7) Due to the advent of patient and reagent barcoding, automated processes within laboratories, the development of high quality instruments with internal quality control mechanisms, and electronic data transfer, diagnostic lab errors occur infrequently—but they do happen. A key principle with point-of-care testing is that when an error is discovered, one should act immediately to identify the root cause of the error, communicate it to both the care team and the patient, and provide education to prevent the same error from happening again. To the credit of the providers in this case—they acted promptly.
The type of pregnancy testing described in this case is CLIA waived. Although the test itself was accurate, the manual method used to enter results into the patient's record is error-prone. Every effort should be made to remove manual steps that introduce risk of human error. While CMS does not require a risk management–based IQCP for such waived tests, this case illustrates that we need safeguards—oversight, protocols, training, and competency assessment—to prevent errors. For some point-of-care tests, proficiency testing will help.(8)
In its early years, point-of-care testing had few controls, but this has changed. The Kost error taxonomy (9,10) and subsequent modifications and adaptations (11-13) introduced features that helped ensure security, performance, and validity throughout the preanalytic, analytic, and postanalytic phases of testing (Figure 2). Instrumented tests incorporated these features into onboard software. Usernames and personal identification numbers became standards for point-of-care testing. All testing should include process features that safeguard results and patient privacy. When dealing with manual tests, such as a pregnancy test, a one-page written plan (i.e., an IQCP) guiding the actions of clinic staff can help ensure that essential safeguards occur.
Ideally, in the outpatient setting, point-of-care testing should be integrated into workflow to minimize errors. The key steps are as follows:
- Ensure that both the provider and the individual performing the test are familiar with necessary testing requirements and manufacturer instructions.
- Design workflow so that the test result is documented immediately upon receipt in the patient's record (paper or electronic). If this workflow is not possible, then identify an appropriate staff member who can achieve this requirement.
- As part of training, include the importance of resulting at the time of testing, not at the end of a shift.
- Make sure the provider is aware (accountability) of the testing requirements for each individual test and patient.
When patients use point-of-care testing at home, such as international normalized ratio for monitoring warfarin anticoagulation therapy or self-testing of blood glucose for diabetes, education and guidance should be provided to the patient in either the hospital or primary care setting. For example, a nurse educator can review the appropriate instructions regarding a home-use glucose meter prior to a patient's discharge from the hospital. The patient's use of point-of-care glucose testing can be reviewed when the patient follows up with his or her primary care physician in the clinic. Communicating detailed information about point-of-care testing to the patient can reduce the potential for preanalytic errors, such as inappropriate sample collection and sample handling. Additionally, if concerns arise about the accuracy of point-of-care pregnancy test results, primary care providers might consider consulting a clinical pathologist for additional testing. Such testing can check for the presence of elements that might confound point-of-care test results, especially in cases of ectopic pregnancy.(14)
From a larger systems viewpoint, a recent study recommended the introduction of a national point-of-care testing guideline, dedicated to primary care and in line with ISO [International Organization for Standardization] standards.(15) Moreover, there has also been discussion about the wisdom of mandatory pregnancy testing before radiological procedures.(16) Review of policy and guidelines for point-of-care testing worldwide (17) supports the hypothesis that a guideline for POC pregnancy testing with sample IQCP templates included would be useful for practitioners and patients alike.
Take-Home Points
- Supposedly simple waived point-of-care testing is neither foolproof nor error free.
- Evaluate available testing methods and devices first, then select those appropriate to the testing situation.
- Start with a plan/protocol (from the ordering of a test to result recording) that ensures test quality, taking an individualized quality control program or similar approach.
- Wherever possible, manual process steps should be eliminated when performing point-of-care testing.
- Once a test error is discovered, correction, communication, and education should follow immediately, accompanied by necessary changes in the testing plan for long-term sustainability.
- Analyst training and competency assessment is essential, though it is not a CLIA requirement for waived testing.
Gerald J. Kost, MD, PhD, MS Director, Point-of-Care Testing Center for Teaching and Research School of Medicine 2016-17 Edward A. Dickson Endowed Emeritus Professor University of California, Davis
Sharon Ehrmeyer, PhD Professor Emeritus, Pathology and Laboratory Medicine School of Medicine and Public Health University of Wisconsin
References
1. Kost GJ. Principles and Practice of Point-of-Care Testing. Philadelphia, PA: Lippincott Williams & Wilkins; 2002. ISBN: 9780781731560.
2. Kost GJ, Curtis CM, eds. Global Point of Care: Strategies for Disasters, Emergencies and Public Health Resilience. Washington, DC: AACC Press; 2014. ISBN: 9781594251726.
3. Howerton D, Anderson N, Bosse D, Granade S, Westbrook G. Good laboratory practices for waived testing sites: survey findings from testing sites holding a certificate of waiver under the Clinical Laboratory Improvement Amendments of 1988 and recommendations for promoting quality testing. MMWR Recomm Rep. 2005;54(RR-13):1-25. [go to PubMed]
4. IQCP: Individualized Quality Control Plan. Developing an IQCP—A Step-by-Step Guide. Baltimore, MD: Centers for Medicare & Medicaid Services. [Available at]
5. Ehrmeyer SS, Laessig RH. Point-of-care testing, medical error, and patient safety: a 2007 assessment. Clin Chem Lab Med. 2007;45:766-773. [go to PubMed]
6. Ehrmeyer SS. Plan for quality to improve patient safety at the point of care. Ann Saudi Med. 2011;31:342-346. [go to PubMed]
7. Ehrmeyer SS. The New Poor Lab's Guide to the Regulations. Madison, WI: Westgard QC, Inc.; 2015. ISBN: 139781886958296.
8. Aslan B, Stemp J, Yip P, Gun-Munro J. Method precision and frequent causes of errors observed in point-of-care glucose testing: a proficiency testing program perspective. Am J Clin Pathol. 2014;142:857-863. [go to PubMed]
9. Kost GJ. Preventing medical errors in point-of-care testing: security, validation, safeguards, and connectivity. Arch Pathol Lab Med. 2001;125:1307-1315. [go to PubMed]
10. Kost GJ. Preventing problems, medical errors, and biohazards in point-of-care testing: using complex adaptive systems to improve outcomes. Point Care. 2003;2:78-88. [Available at]
11. Jones BA, Meier FA. Patient safety in point-of-care testing. Clin Lab Med. 2004;24:997-1022. [go to PubMed]
12. Meier FA, Jones BA. Point-of-care testing error: sources and amplifiers, taxonomy, prevention strategies, and detection monitors. Arch Pathol Lab Med. 2005;129:1262-1267. [go to PubMed]
13. Plebani M. Does POCT reduce the risk of error in laboratory testing? Clin Chim Acta. 2009;404:59-64. [go to PubMed]
14. Kurt-Mangold M, Van Voorhis BJ, Krasowski MD. Persistent human chorionic gonadotropin after methotrexate treatment and an emergency surgical procedure for ectopic pregnancy. Lab Med. 2015;46:254-258. [go to PubMed]
15. de Vries C, Doggen C, Hilbers E, et al. Results of a survey among GP practices on how they manage patient safety aspects related to point-of-care testing in every day practice. BMC Fam Pract. 2015;16:9. [go to PubMed]
16. Applegate K. Pregnancy screening of adolescents and women before radiologic testing: does radiology need a national guideline? J Am Coll Radiol. 2007;4:533-536. [go to PubMed]
17. Kost GJ, bt Mohd Hussain B. National point of care testing policy and guidelines in Malaysia, standards of care, and impact worldwide. In: Kost GJ, Curtis CM, eds. Global Point of Care: Strategies for Disasters, Emergencies and Public Health Resilience. Washington, DC: AACC Press; 2014:595-610. ISBN: 9781594251726.
Figures
Figure 1. The Conceptual Basis and Process Steps of an Individualized Quality Control Plan.
Figure 2. Schematic Illustrating Elements of the Modified Kost Taxonomy for Reducing Errors in Point-of-care Testing.