Amended Lab Results: Communication Slip
Mohta V. Amended Lab Results: Communication Slip. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2012.
Mohta V. Amended Lab Results: Communication Slip. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2012.
The Case
A 25-year-old woman in her first pregnancy was seen at 33 weeks' gestation with new onset hypertension and a blood pressure of 140–150/90 mm Hg. Urinalysis revealed 2+ protein (100 mg/dL) on a random urine dipstick, and a urine protein/creatinine ratio was 0.5 mg/dL (normal up to 0.3 mg/dL). With a potential diagnosis of preeclampsia (new onset hypertension and proteinuria during pregnancy), she was admitted to the hospital for observation, fetal testing, and laboratory studies, including a 24-hour urine protein collection. In the absence of proteinuria, this syndrome is considered gestational hypertension, a less serious diagnosis, managed as an outpatient with either observation or antihypertensive medications.
The laboratory studies and 24-hour urine collection for proteinuria were checked by the obstetrics/gynecology (OB/GYN) resident in the hospital electronic medical record (EMR), where they were reported as normal. Assuming a diagnosis of gestational hypertension, the resident initiated discharge planning, including management with antihypertensive medications. However, the attending physician, noting the inconsistency between the dipstick proteinuria and negative result on the 24-hour urine, rechecked the laboratory results. Upon recheck, he observed that the laboratory had amended the report confirming the 24-hour protein collection was actually elevated and abnormal, confirming the diagnosis of preeclampsia. The patient's discharge was canceled.
In this instance, the amended laboratory result was recognized prior to discharge and a new management plan, which involved continued observation in the hospital, was implemented. No harm was done to the patient. She remained in the hospital for 5 days and was induced at 34 weeks for worsening preeclampsia. The mother and infant ultimately did well.
On later review, it was determined that the nature of this error centered upon the communication of an amended test or report. Although this hospital's policy mandated that the inpatient unit be notified about amended laboratory reports, notifying the physician was not part of the mandate. Consequently, the ordering physician was not alerted to the change in the laboratory report.
The Commentary
This case highlights two medical errors, each with important causes and consequences.
The first was a laboratory error. The laboratory in this case initially reported an incorrect value for the 24-hour urine. Because hospitals process thousands of samples per day, some amended results are inevitable, even though error rates are low (around 0.3%).(1) As in other arenas of medicine, most erroneous laboratory reports are the result of human error, ranging from labeling errors to transcription errors. Errors of the assay process are much less common.(2) Many laboratory errors are of no clinical consequence. However, approximately 30% of amended laboratory values do lead to a change in patient care (3,4), as seen in this case.
The second error was a failure to communicate this changed value to the responsible clinician. Failure of communication between the laboratory and the clinician is a well-known and frequent cause of medical error. As is made clear by this case, notifying the inpatient unit is often insufficient. Even when the laboratory staff attempts to notify a physician rather than the floor, in teaching hospitals frequent resident turnover complicates identification of the responsible clinician. Often the resident who ordered the original test is no longer in the hospital.
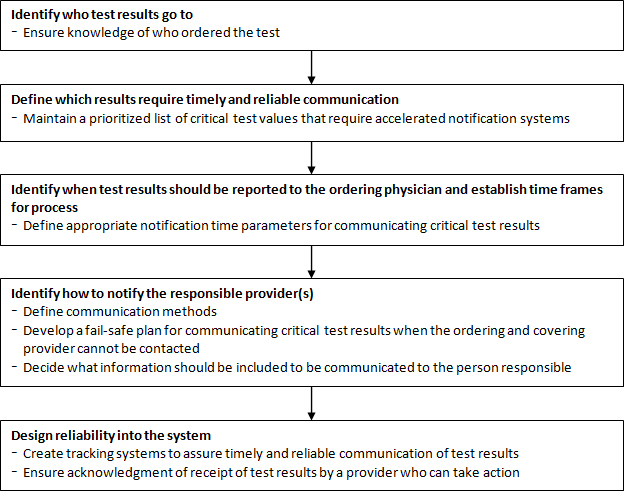
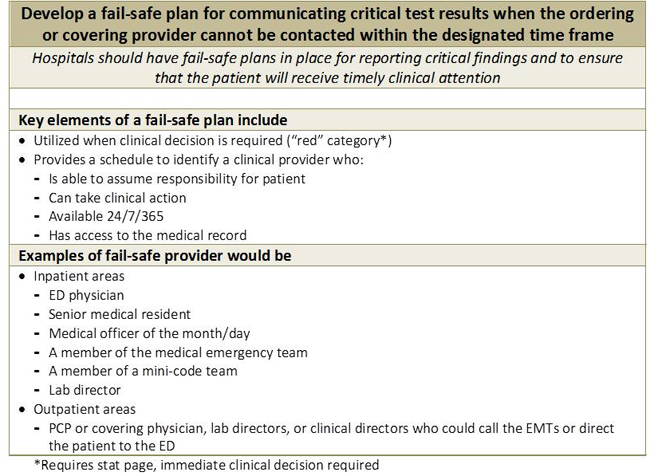
Several groups have developed best practice guidelines for communicating critical test results (Figure 1).(5,6) A critical aspect of lab communication is clear identification of a responding clinician 24/7 who can take clinical action. In the inpatient setting, the responsibility lies with hospitals to develop such a method of identification.(6) Creating a fail-safe system of communication between two silos, the clinical team and the laboratory team, is difficult and requires significant effort on both sides. A critical component of the communication is an escalation list of who should be called until a responsible physician is reached and a time frame for each step (Figure 2).(6)
Communicating an amended value may be even more difficult than communicating a critical value. With amended values, the clinician is not anticipating new results and may have already acted on erroneous information. Despite these challenges, there are few, if any, standard-of-care guidelines to assist hospitals in designing processes of communication of amended values. This case shows us that the communication must be immediate and proactive. The laboratory personnel member who amends the result is the logical person to be directly responsible for communication with a responding clinician as defined above. The algorithm should call for continued efforts by the laboratory until this goal has been achieved, similar to communication of critical results. A tracking system can help to ensure integrity of the process.
In this specific case, it is possible that the laboratory did not try to communicate with a clinician with more urgency because the final value of the 24-hour urine protein was not on a list of critical values. However, defining critical values can be problematic, particularly in obstetrics. The laboratory is often unaware that the patient is pregnant. Values that may be normal outside of pregnancy can be cause for concern in pregnancy. Alternatively, a critical value for an internist may appear to be normal to an obstetrician. For example, a creatinine of 1.2 mg/dL will raise an obstetrician's eyebrows, but a D-dimer of 600 μg/L will not.
In this case, the diagnosis in question was preeclampsia. Preeclampsia is diagnosed by elevated blood pressure (consistently greater than 140/90 mm Hg) and proteinuria (greater than 300 mg in 24 hours). Preeclampsia is associated with high maternal and fetal risks, often leads to prematurity, and in most cases demands inpatient management; furthermore, there is a big difference between 250 mg and 350 mg of proteinuria. In this case, it is unlikely that the laboratory appreciated that 300 mg was an important cutoff for the clinicians.
Finally, in this case, patient harm was avoided by a senior clinician noticing a discrepancy between the urine dipstick and the 24-hour urine results. Could an automated system within the laboratory or within the electronic medical record have flagged these discrepant results and triggered a double-check of the 24-hour urine result prior to reporting? Some commercial labs perform comparisons of current and prior values, and double check results that show a large change.(7) An EMR that could perform such complex comparisons would require significant infrastructure to design and maintain, and many practical obstacles would need to be overcome. For now, there is no electronic replacement for an astute clinician who can compare and trend values. However these complex systems may have some promise for the future. If housed within the EMR, such systems may even be able to tailor critical or discrepant values to the patient's condition and reason for ordering the test (pregnancy, sepsis, etc.), further reducing errors and improving communication. Continued development of these software systems may play a key role in reducing the type of errors noted in this case in the decade to come.
Take-Home Points
- Amended laboratory results can result in patient care errors because clinicians are not anticipating a change in information.
- Clinical laboratories should follow strict algorithms for contacting a responsible clinician when communicating amended results, similar to the ones they follow when communicating critical values.
- Automated systems to recognize critical values function less well in the obstetrics setting or other settings in which baseline values may be atypical.
- While not currently in wide adoption, smart systems of information technology that identify errors and critical values may have promise for the future.
Vanitha Janakiraman Mohta, MD, MPH Maternal Fetal Medicine Kaiser Permanente Medical Group
References
1. Carraro P, Plebani M. Errors in a stat laboratory: types and frequencies 10 years later. Clin Chem. 2007;53:1338-1342. [go to PubMed]
2. Nutting PA, Main DS, Fischer PM, et al. Toward optimal laboratory use. Problems in laboratory testing in primary care. JAMA. 1996;275:635-639. [go to PubMed]
3. Howanitz PJ. Errors in laboratory medicine: practical lessons to improve patient safety. Arch Pathol Lab Med. 2005;129:1252-1261. [go to PubMed]
4. Astion ML, Shojania KG, Hamill TR, Kim S, Ng VL. Classifying laboratory incident reports to identify problems that jeopardize patient safety. Am J Clin Pathol. 2003;120:18-26. [go to PubMed]
5. Gandhi TK. Fumbled handoffs: one dropped ball after another. Ann Intern Med. 2005;142:352-358. [go to PubMed]
6. Hanna D, Griswold P, Leape LL, Bates DW. Communicating critical test results: safe practice recommendations. Jt Comm J Qual Patient Saf. 2005;31:68-80. [go to PubMed]
7. Bates DB, Cohen M, Leape LL, Overhage JM, Shabot MM, Sheridan T. Reducing the frequency of errors in medicine using information technology. J Am Med Inform Assoc. 2001;8:299-308. [go to PubMed]
Figures
Figure 1. Communicating critical test results: Creating a fail-safe process.(5)
© American College of Physicians: Gandhi TK. Fumbled handoffs: one dropped ball after another. Ann Intern Med. 2005;142:352-358. Reprinted with permission.
Figure 2. Developing a fail-safe communication system.(6)
© Joint Commission Resources: Hanna D, Griswold P, Leape LL, Bates DW. Communicating critical test results: safe practice recommendations. Jt Comm J Qual Patient Saf. 2005;31:68-80. Reprinted with permission.