Pathologic Mistake
The Case
A 32-year-old previously healthy woman experienced abdominal pain and bloating for 6 months. The discomfort worsened with eating. After losing 15 pounds in less than 2 months, she visited her primary care physician. Initial imaging showed enlarged abdominal lymph nodes as well as jejunal thickening and dilated ileum in the small bowel. A lymph node biopsy and partial small bowel resection was undertaken. Lymph node pathology was reported as "Castleman disease, of hyaline vascular type," while the small bowel biopsy was normal.
Castleman is a rare disease in which immune cells in the lymph nodes proliferate. It is not considered a cancer, but is treated by physicians who specialize in lymphomas. With this diagnosis, her primary physician appropriately referred her to the lymphoma clinic. Physical exam, laboratory tests, and repeat computed tomography (CT) scans were all consistent with the initial workup, and the specialist began making plans to administer chemotherapy for Castleman. Still, the lymphoma specialist was struck by a number of inconsistencies. The combination of the location and number of affected lymph nodes would have made this an extremely rare presentation of an already rare disease. Also, CT findings of jejunal thickening and a dilated ileum were atypical for this diagnosis.
Puzzled, the lymphoma specialist requested a second pathology opinion from a tertiary care center with expertise in Castleman disease. The review reported reactive lymph nodes with hyalinization but no other features of Castleman, and normal small bowel. Plans for chemotherapy were cancelled. Lacking a diagnosis, further work-up was initiated. Upper gastrointestinal endoscopy and blood work was consistent with adult-onset celiac disease. She was started on a gluten-free diet. Follow-up imaging at 4 months showed dramatic improvement, and with strict adherence to her diet, the patient is still symptom-free.
The Commentary
This case highlights how diagnostic pathology errors can lead to incorrect patient management plans. In this commentary, we will categorize anatomic pathology errors and discuss prevention strategies. In this case, the error is an incorrect diagnosis of Castleman disease, a rare disorder in which lymph node tissue grows at an abnormally fast rate. Occurring most commonly in adults (mainly younger than 30 years), it is benign and is often confused with other similar benign or malignant conditions, such as reactive lymph nodes and lymphomas.(1-4)
Anatomic pathology is prone to error because the testing process has many complex steps. Interestingly, this makes it a particularly fertile specialty for a more structured approach to error detection and prevention.(5-10) The key steps in pathology laboratory workflow include obtaining a tissue sample (specimen procurement), patient and specimen identification, and then specimen transport to the pathology laboratory. Once the specimen is processed, an attending pathologist interprets the specimen and reports the results, which the ordering clinician(s) receives. Each of these steps carries the possibility of error.
While there are many different steps in the pathology process, they can be organized into three distinct phases (6):
- i. Preanalytic phase: clinician test selection, test ordering, specimen procurement, patient and specimen identification, and specimen transport;
- ii. Analytic phase: specimen processing, preparation, and assessment by the pathologist with immediate reporting of test results;
- iii. Postanalytic phase: receipt of the test results by the clinician followed by clinician interpretation of the test results, and clinical action based on that interpretation.
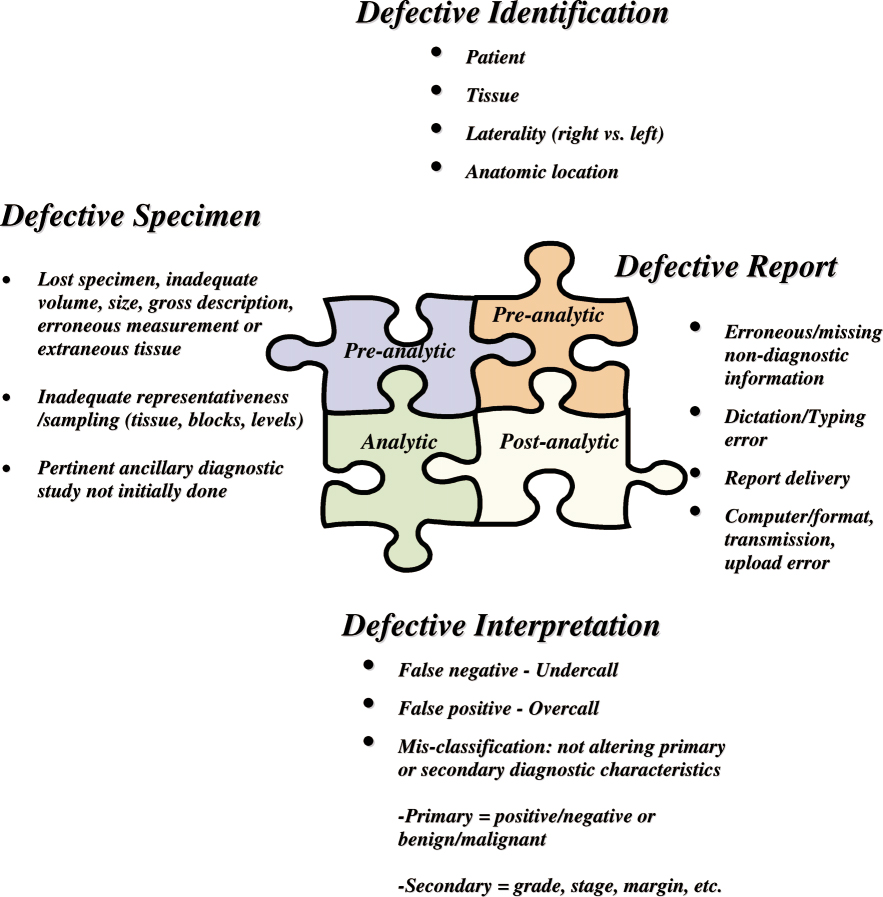
Zarbo and colleagues (6) proposed four error types in anatomic pathology: identification, specimen, report, and interpretation (Figure). The authors further divided interpretation errors into three subcategories: (i) false-negative diagnosis (undercall of the extent or severity of a lesion), (ii) false-positive diagnosis (overcall of the extent or severity of a lesion), and (iii) misclassification.(6) In the present case, the error stems from a defective interpretation with an overcall diagnosis. Broadening our discussion to error theory in general, errors related to knowledge-based cognitive behaviors occur when the person is unable to apply his or her existing knowledge to a novel situation. This kind of error can be difficult to identify because it can be confused with other error types, such as human lapses.(6)
Root cause analyses of diagnostic errors, such as the false-positive diagnosis of Castleman disease in this case, generally reveal both active and latent errors. Lack of training, high workload volume, and interruptions are examples of latent conditions that increase the likelihood of error. Latent errors also include insufficiently redundant system checks, and failures in preanalytic (e.g., obtaining less than an optimal specimen sample or inadequate clinical history that compromises diagnostic interpretation) and analytic steps. Active errors include heuristic biases related to diagnostic decision making and ancillary test ordering. For instance, the tendency of a pathologist to lock onto a diagnosis (usually early in the workup of a case) and then undervalue data that would either support another diagnosis or refute the favored diagnosis represents an active error (and an example of an anchoring heuristic).
The error classification system above was proposed to improve measurement of error rates, clinical impact of errors, and the effect of error reduction and prevention efforts.(6) Studies in anatomic pathology, which have shown overall error rates of about 5% have estimated these rates by phase: preanalytic phase (2.0%), analytic phase (0.2%), and postanalytic phase (2.2%). The error rates in pathology also vary by the type of review. For example, pathology errors in cancer diagnosis are relatively common and can have high impact on the patient.(11) Such errors (defined as discordance between cytologic and histologic interpretation) occur in up to 11.8% of all reviewed cytologic-histologic specimen pairs. Yet, disagreement often exists on whether a given pathology error was secondary to misinterpretation or poor clinical sampling of tissues.(9)
A number of factors affect the severity of harm associated with pathology interpretation error. In assessing the clinical impact of errors detected by cytologic-histologic case correlation, one study reported that in non-gynecologic specimens 11.7% to 100% of detected errors caused no harm, while another institution reported that 4.9% of detected errors led to severe harm.(9) The variability of these results is partly a function of anatomic site (e.g., bone versus lymph node), but also is due to the inadequate taxonomies and assessment processes currently used for classifying error severity. Raab and colleagues (9) recommended an error severity scale based on several factors such as morbidity, mortality, delay in diagnosis, and additional testing performed. They also reported that pathologists disagreed on the extent of harm caused by diagnostic errors. In most areas of anatomic pathology, if harm occurs, the severity is generally mild or minimal (depending on the classification system) and results in additional testing, delays, and/or patient anxiety.(8) For instance, a common benign skin lesion, seborrheic keratosis, could mistakenly be diagnosed as acrochordon (skin tag), which also is a benign skin lesion, causing no harm and requiring the same management. In contrast, when the same skin lesion is erroneously diagnosed as squamous cell carcinoma (a skin cancer requiring different management), the impact of the error is more serious.
Error prevention requires a multifaceted approach. Preventive strategies such as double-reading (by two separate pathologists), case conference review (i.e., discussion of histology results between pathologist and clinician) prior to sign-out, and improved use of consultation (second opinion) are all well practiced and recognized in anatomic pathology. The interpretation error in the case described above could have well been prevented with such strategies as double-reading or second opinion. Marked differences in both definitions of error and pathology practice make it impossible to compare the efficacy of published error detection and prevention interventions.(6) Studies have suggested that observer redundancy (e.g., directed slide review of particular case types, random review of a fixed percentage of cases, ad hoc consultation with other pathologists, and presentation of "difficult" cases at daily conferences) reduces diagnostic variation and interpretive error, which ranges from 1.2 to 50 errors per 1000 cases. However, it is unclear which forms of such redundancy are the most effective in uncovering diagnostic error. Some hospitals require second interpretations by local pathologists in specified situations such as a new cancer diagnosis. Additionally, using root cause analysis to identify sources of error has proven to decrease error.(11)
Clinicians can also play an essential role in pathology error reduction through several avenues such as appropriate test ordering, dispensing accurate and pertinent clinical information, procuring high-quality specimens, providing timely follow-up on test results, effectively communicating (with pathology) on potentially discrepant diagnoses, and advocating second opinions in specific situations (e.g., discordance between clinical impression and pathology diagnosis).(8,12) Procurement of high-quality specimens is enhanced when pathology is required to provide an immediate interpretation of fine needle aspirations (FNA), thereby facilitating resampling by the clinician when necessary. Strategies to promote these behaviors include training in and standardization of pathologist–clinician communication to facilitate inclusion of all important data elements and reduce ambiguity in terminology.
Take-Home Points
- Anatomic pathology is prone to error at many steps due to the complex nature of the testing process.
- Error rates in anatomical pathology are relatively low. Although some errors have little clinical consequence, others may be more severe.
- Interpretation errors are important and often difficult to understand and classify. Efforts to develop a classification scheme will help rationalize the area of pathology patient safety.
- Preventive strategies such as double-reading, case conference review, and second opinion (mandatory in specified situations) can reduce interpretation errors. In addition, standardizing pathologist–clinician communication and requiring immediate interpretation of all FNA samples are examples of strategies that institutions have adopted to reduce pathology-related errors.
Reza Alaghehbandan, MD, MSc Resident, Anatomic Pathology Memorial University of Newfoundland St. John's, NL, Canada
Stephen S. Raab, MD Professor, Anatomic Pathology Memorial University of Newfoundland Clinical Chief, Laboratory Medicine, Eastern Health Authority St. John's, NL, Canada
References
1. Ghosh A, Pradhan SV, Talwar OP. Castleman's disease—hyaline vascular type—clinical, cytological and histological features with review of literature. Indian J Pathol Microbiol. 2010;53:244-247. [go to PubMed]
2. Zarate-Osorno A, Medeiros LJ, Danon AD, Neiman RS. Hodgkin's disease with coexistent Castleman-like histologic features. A report of three cases. Arch Pathol Lab Med. 1994;118:270-274. [go to PubMed]
3. Menke DM, Tiemann M, Camoriano JK, et al. Diagnosis of Castleman's disease by identification of an immunophenotypically aberrant population of mantle zone B lymphocytes in paraffin-embedded lymph node biopsies. Am J Clin Pathol. 1996;105:268-276. [go to PubMed]
4. Siddiqi IN, Brynes RK, Wang E. B-cell lymphoma with hyaline vascular Castleman disease-like features: a clinicopathologic study. Am J Clin Pathol. 2011;135:901-914. [go to PubMed]
5. Sirota RL. Defining error in anatomic pathology. Arch Pathol Lab Med. 2006;130:604-606. [go to PubMed]
6. Zarbo RJ, Meier FA, Raab SS. Error detection in anatomic pathology. Arch Pathol Lab Med. 2005;129:1237-1245. [go to PubMed]
7. Meier FA, Zarbo RJ, Varney RC, et al. Amended reports: development and validation of a taxonomy of defects. Am J Clin Pathol. 2008;130:238-246. [go to PubMed]
8. Raab SS, Grzybicki DM. Quality in cancer diagnosis. CA Cancer J Clin. 2010;60:139-165. [go to PubMed]
9. Raab SS, Grzybicki DM, Janosky JE, et al. Clinical impact and frequency of anatomic pathology errors in cancer diagnoses. Cancer. 2005;104:2205-2213. [go to PubMed]
10. Raab SS, Nakhleh RE, Ruby SG. Patient safety in anatomic pathology: measuring discrepancy frequencies and causes. Arch Pathol Lab Med. 2005;129:459-466. [go to PubMed]
11. Raab SS, Grzybicki DM, Zarbo RJ, Meier FA, Geyer SJ, Jensen C. Anatomic pathology databases and patient safety. Arch Pathol Lab Med. 2005;129:1246-1251. [go to PubMed]
12. Raab SS, Meier FA, Zarbo RJ, et al. The "Big Dog" effect: variability assessing the causes of error in diagnoses of patients with lung cancer. J Clin Oncol. 2006;24:2808-2814. [go to PubMed]
Figure
Figure. Error types in anatomic pathology. Reprinted from (6) with permission from the College of American Pathologists.