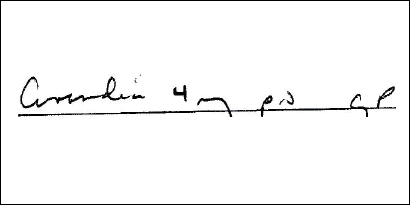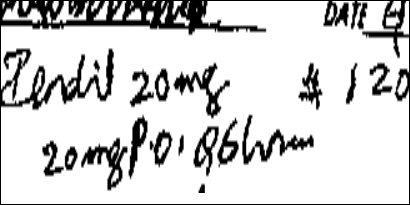Medication Overdose
The Case
A 15-year-old boy with end-stage AIDS was admitted to the pediatric ICU with mental status changes. He was diagnosed with status epilepticus and started on a loading dose of IV phenytoin.
In the step-down unit, the resident wrote an order for a maintenance dose of phenytoin. The order was written as mg/kg/d without specification that ’d’ meant day vs. dose. As a result, the patient received approximately three times the indicated dose. Later that day, a pharmacist called to alert the resident to his mistake. The subsequent phenytoin level was 98 (therapeutic range 10-20).
Administration of phenytoin was held until the level was therapeutic, and the patient’s mental status gradually improved. He had no further seizure activity and ultimately his mental status returned to baseline. He was discharged back to a chronic care facility.
The Commentary
The case at hand may strike many readers as uncommon or unlikely to befall careful and competent providers, but errors like this are frequent and may have severe sequelae. In fact, one of the most publicized medical errors in recent years involved a very similar fatal misinterpretation of a dosing abbreviation. In 1994, Boston Globe health columnist Betsy Lehman died after receiving a four-fold overdose of cyclophosphamide.(1) The ordering physician had written “cyclophosphamide 4g/sq m over four days” with the intention that the patient receive a total of 1 gram per square meter over 4 sequential days. Unfortunately, the order was interpreted as prescribing 4 grams per square meter for each of 4 sequential days. Lehman died 3 weeks later from the cardiac complications ensuing from this massive overdose. Lest readers think this error represents a tragic “fluke,” another breast cancer patient received an identical overdose (though non-fatal) around the same time.(1) Why do these errors occur and what can we do to prevent them?
In large studies of hospitalized adults, medication errors represent the most frequent cause of injuries from medical care.(2,3) Roughly 5% of hospitalized adults experience an adverse drug event (ADE), and an additional 5% experience a potential ADE (ie, the error reached the patient, but no harm occurred).(4) A recent study from an academic children’s hospital reported similar rates of ADEs, though rates of potential ADEs were even higher.(5) Most potential ADEs occurred at the stage of drug ordering (79%) and involved incorrect dosing in 34% of these cases.(5)
Errors in drug ordering include errors in drug choice (eg, failure to note an important drug–drug interaction or a relevant allergy) and errors in dosing. Related to drug ordering is prescription transcription, as accurately conveying the drug choice or dose are clearly crucial steps in the medication process. Physicians’ handwriting has become a perennial joke. Although one study suggested that physicians’ handwriting is no more illegible than that of lawyers and businessmen,(6) physicians’ handwriting, when illegible, can have life-threatening consequences.(Figure 1, Figure 2)
The error in this case occurred independent of prescription legibility. The confusion between ‘dose’ and ‘day,’ due to ambiguous use of the abbreviation “d” in “mg/kg/d,” could have occurred under many possible circumstances. Another common context for this type of confusion is an order such as “phenytoin 100mg po q8h x 3d”—does this mean for 3 days or doses?
Dosing errors are common in adults and children,(5,7) but “overdose errors” may be particularly likely in pediatrics.(8,9) Doses are often derived from weight-based calculations, and physicians frequently err when performing these calculations.(10) In the pharmacy, solution volumes tend to be small, so large overdoses—even the tenfold errors that can occur from erroneous calculations or misplaced decimals—may not look suspicious.(9)
In this case, the patient developed a high serum phenytoin level, but experienced no clinical toxicity, so the event would be counted as a potential ADE. The lack of clinical consequences to the error reflected the pharmacist’s recognition of the ambiguous order before more than one dose had been administered. Pharmacists are often our first line of defense against medication errors, and ward-based clinical pharmacists can be particularly valuable by reducing ADEs through ongoing monitoring and correction of orders prior to processing.(11)
In addition to involving pharmacists more directly in the ordering process, prevention strategies can include educational interventions directed at physicians, targeting abbreviations and other prescribing practices (8) and even handwriting legibility.(12) However, more lasting and reliable solutions are clearly desirable.
Computerized physician order entry (CPOE) is the most effective intervention to eliminate the type of error presented in this case.(13) CPOE refers to a software application that allows doctors to electronically write all orders and replaces handwritten orders. Even the most basic CPOE system eliminates issues of illegibility and ensures that orders are complete and in a standard format, eliminating ambiguous dosing orders as occurred in this case. More sophisticated CPOE systems provide additional “decision support,” such as warnings about relevant allergies or drug interactions or suggested dose changes based on recent lab values (eg, “renal dosing” based on most recent serum creatinine).(14)
Take-Home Points
Since most hospitals do not have such sophisticated medication ordering systems, the need remains for physicians to observe basic patient safety practices related to order writing. These include:
- Always write orders legibly.
- Use abbreviations sparingly.(Table)
- Encourage institutional development of approved abbreviations.
- Communicate directly to nurses and pharmacists regarding potentially confusing orders.
- Encourage institutional employment of information technologies, such as CPOE.
- Encourage institutional employment of other systems-based error prevention strategies such as employing ward-based clinical pharmacists.
Rainu Kaushal, MD, MPH Departments of Medicine and Pediatrics Brigham and Women’s Hospital Harvard Medical School
References
1. Knox RA. Doctor's orders killed cancer patient. Boston Globe. March 23, 1995.
2. Brennan TA, Leape LL, Laird NM, et al. Incidence of adverse events and negligence in hospitalized patients. Results of the Harvard Medical Practice Study I. N Engl J Med. 1991;324:370-376.[ go to PubMed ]
3. Thomas EJ, Studdert DM, Burstin HR, et al. Incidence and types of adverse events and negligent care in Utah and Colorado. Med Care. 2000;38:261-271.[ go to PubMed ]
4. Bates DW, Cullen DJ, Laird N, et al. Incidence of adverse drug events and potential adverse drug events. Implications for prevention. ADE Prevention Study Group. JAMA. 1995;274:29-34.[ go to PubMed ]
5. Kaushal R, Bates DW, Landrigan C, et al. Medication errors and adverse drug events in pediatric inpatients. JAMA. 2001;285:2114-2120.[ go to PubMed ]
6. Berwick DM, Winickoff DE. The truth about doctors' handwriting: a prospective study. BMJ. 1996;313:1657-1658.[ go to PubMed ]
7. Lesar TS, Lomaestro BM, Pohl H. Medication-prescribing errors in a teaching hospital. A 9-year experience. Arch Intern Med. 1997;157:1569-1576.[ go to PubMed ]
8. Koren G. Trends of medication errors in hospitalized children. J Clin Pharmacol. 2002;42:707-710.[ go to PubMed ]
9. Koren G, Barzilay Z, Greenwald M. Tenfold errors in administration of drug doses: a neglected iatrogenic disease in pediatrics. Pediatrics. 1986;77:848-849.[ go to PubMed ]
10. Lesar TS. Errors in the use of medication dosage equations. Arch Pediatr Adolesc Med. 1998;152:340-344.[ go to PubMed ]
11. Leape LL, Cullen DJ, Clapp MD, et al. Pharmacist participation on physician rounds and adverse drug events in the intensive care unit. JAMA. 1999;282:267-270.[ go to PubMed ]
12. Shaughnessy AF, D'Amico F. Long-term experience with a program to improve prescription-writing skills. Fam Med. 1994;26:168-171.[ go to PubMed ]
13. Bates DW, Leape LL, Cullen DJ, et al. Effect of computerized physician order entry and a team intervention on prevention of serious medication errors. JAMA. 1998;280:1311-1316.[ go to PubMed ]
14. Chertow GM, Lee J, Kuperman GJ, et al. Guided medication dosing for inpatients with renal insufficiency. JAMA. 2001;286:2839-2844.[ go to PubMed ]
Table
Table. Prescription Shorthand that May Lead to Errors
|
Abbreviation or Prescribing Practice |
Error |
Recommendation |
|---|---|---|
|
µg for microgram |
Misread as mg for milligram |
Use ‘mcg’ |
|
U for units |
Messy U easily confused for zero – eg, Insulin 10U becomes Insulin 100 |
Write out ‘units’ |
|
Abbreviations of drug names |
Multiple possible name confusions – eg, HCTZ (hydrochlorothiazide) misinterpreted as HCT (hydrocortisone) or CPZ (Compazine or chlorpromazine?); MSO4/MgSO4 for morphine and magnesium sulfate, respectively |
Spell out name in full |
|
Trailing zero after decimal point (e.g. 1.0) |
Misread as 10 instead of 1 if the decimal point is not seen |
Do not use terminal zeroes for doses (1 mg, not 1.0 mg, but 0.5 mg, not .5 mg) |
Figures
Figure 1. Avandia® Confused With Coumadin®
A poorly written order for the diabetic medication, AVANDIA (rosiglitazone), bears a strong resemblance to the oral anticoagulant, COUMADIN (warfarin). The potential for this potentially disastrous medication substitution is accentuated by the fact that both drugs are available as 4 mg oral tablets.
Figure 2. Plendil®, Confused With Isordil®
This poorly written prescription for for Isordil® (isosorbide dinitrate) 20 mg q 6 hours was interpreted by the pharmacist and dispensed as Plendil® (felodipine) 20 mg q 6 hours. The patient suffered a myocardial infarction after only one day of taking the erroneous prescription; he died a few days later. The pharmacist and physician were both sued in this case – the physician for the illegible handwriting itself and the pharmacist for not questioning the illegible prescription, especially given that the interpreted order far exceeded the recommended maximum dose of Plendil (10 mg daily).