Unexplained Apnea Under Anesthesia
Barach P. Unexplained Apnea Under Anesthesia. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2003.
Barach P. Unexplained Apnea Under Anesthesia. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2003.
Case Objectives
Clinical Objectives
- List the causes of prolonged apnea in the operating room
- Describe the steps in management of apnea in the operating room
Patient Safety Objectives
- State the prevalence of medication errors
- List the causes of wrong drug administration in the operating room
- Describe the system checks available to prevent medication errors in the operating room
Case & Commentary: Part 1
A 15-year-old boy underwent elective right knee arthroscopy and debridement under general anesthesia with a laryngeal mask airway (LMA). He was otherwise healthy with no allergies to medications. After uneventful induction of anesthesia, the surgeons requested antibiotic prophylaxis with cefazolin 1 gram, which the anesthesiology team administered. Just before the surgical incision was made, 50 mcg of Fentanyl was administered. About 2 minutes later, spontaneous respirations slowed, and the patient became apneic. The surgeon and anesthesiologist assumed the patient’s apnea was due to opiate sensitivity and assisted ventilation by hand for 30 minutes. However, despite a rise in the end-tidal CO2 to 70mm Hg, spontaneous respirations did not return.
Apnea during anesthesia has several etiologies, including anesthetic agents themselves, as well as opiates, barbiturates, or benzodiazepines, and hypocarbia-induced respiratory depression. Prolonged apnea occurs more often in hyperventilated patients; neonates; elderly patients; patients with compromised renal, pulmonary, or hepatic function; hypothermic and acidotic patients; patients receiving neuromuscular blockade, aminoglycosides, or intravenous magnesium; and patients with neurological impairment or injury.
Assuming this patient is healthy, normothermic, and not acidotic or hypocarbic, and assuming he did not receive neuroaxial anesthetic blockade (such as spinal or epidural regional anesthesia), I would be concerned that he received an unplanned drug due to a syringe or an ampoule swap.(Table 1) While maintaining cardiovascular and respiratory functions, clinicians should attempt to ascertain whether a wrong drug was administered, and if so, which drug.(Table 2) (1) The most common drugs that may lead to apnea include muscle relaxants or highly potent opiates (such as Sufentanil, which is ten times as potent as fentanyl). Alternatively, the patient may have a previously unrecognized metabolic disorder such as a neuromuscular disease (ie, myasthenia gravis) or a structural abnormality (ie, stroke or embolism) that needs to be evaluated.
Treatment of medication-induced respiratory depression varies by cause. When respiration is depressed by opiates, as evidenced by miotic, unresponsive pupils, naloxone (Narcan) in 0.04 mg increments may be titrated to reverse the condition. In the case of persistent peripheral muscle blockade, typically due to residual muscle relaxants, reversal with neostigmine is initiated. Other interventions include discontinuation of anesthetics, determination of arterial blood gases, and appropriate adjustment of ventilation.(Table 3)
Case & Commentary: Part 2
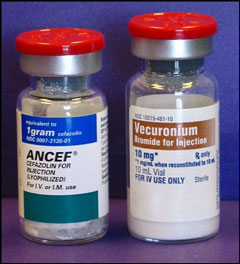
Because the apneic episode lasted longer than 30 minutes, the anesthesia team began to question their initial assumption that the apnea was due to opiate sensitivity. They had obtained the cefazolin from the medication drawer of the anesthesia cart. The anesthesia team examined the drawer and found vials of cefazolin and vecuronium (a long-acting paralytic agent) in adjacent medication slots. The vials were of the same size and shape, with similar red plastic caps.(Figure 1) The team realized that the patient had received vecuronium 10 mg, not cefazolin 1 g, and that the observed apnea was therefore due to unrecognized muscle relaxation.
There is no accurate measure of the morbidity and mortality associated with anesthesia. It has been estimated that between 2000 and 10,000 patients die each year from causes at least partially related to anesthesia, but those estimates are based on circumstantial data and include all patients regardless of age or physical status.(2) A recent study in the United Kingdom found that only 1 patient in 185,000 died solely as result of anesthesia, although 1 in 1351 deaths was in part related to anesthesia.(3) An estimated 44,000-98,000 Americans die in hospitals each year as a result of preventable medical errors.(4) Medication errors were the number one cause of adverse and preventable patient events, leading to more than 7000 deaths annually.(4)
The prevalence of medication errors in the operating room is not accurately known, although it is probably similar to that of medication errors in the hospital. Bates and colleagues have shown that 6.5% of admitted patients suffered an adverse drug event.(5) Of these events, 28% were due to errors, and an additional 5.5% involved near misses caught due to interception of the error. In the Harvard Medical Practice Study, adverse drug events accounted for 19.4% of all disabling adverse events, 45% of those events were caused by errors.(6) In a large insurer’s study, injuries due to drugs were the most frequent cause of procedure-related malpractice claims.(7)
Wrong medication administration in the operating room is due to failure to label syringes, incorrect matching of labels on syringes and drug ampoules, failure to read the label on the vial/ampoule, misuse of decimal points and zeroes, and inappropriate abbreviations. What happened to this patient illustrates an example of faulty drug identity checking, where two drugs were packaged in similar vials, so that one was easily mistaken for the other. Poor system design also makes errors difficult to intercept before injury occurs. Leape and colleagues discovered that failures at the system level were the real culprits in more than three-fourths of adverse drug events.(8) Reason and colleagues suggested that some complex healthcare systems are more vulnerable and therefore more likely to experience adverse events.(9)
Documenting errors at the administration stage is difficult, because it requires direct observations and reliable, robust near-miss and adverse-event reporting systems. Currie and colleagues found 144 incidents related to drugs, of which 58 were related to syringe or drug swaps,(10) among the first 2000 incidents of the Australian Incident Monitoring System. Of those 58 events, 71% involved muscle relaxants. Implementing a red syringe color change for all Succinylcholine drug administration in Australia has helped to reduce drug and syringe swap by 70%.(11) A large, retrospective study of anesthesiologists’ self-reported incidents found that of a total of 1089 incidents, 71 were related to either syringe or drug ampoule swap (7%).(12) Leape and colleagues found that 40 of 334 errors (12%) at the stage of drug ordering and delivery were due to imperfect dose and identify checking.(8) Studies in intensive care units have produced similar results.(13)
Administrative medication errors in the operating room and intensive care unit are believed to be more common in unfamiliar settings, when drug packaging or ampoules have changed, when similarly appearing ampoules are stored close together in the medication carts, when syringes are prepared by other personnel, when hand written labels are used, and when lighting conditions are poor.(14) There is an exponential relationship between the number of drugs administered to a patient and the prevalence of adverse drug events.(15)
Although there is no excuse for failing to read medication and syringe labels, the occasional failure to do so represents an expected “slip,” more likely to occur with fatigue, distraction, or other causes of momentary lapses in concentration and failures in automatic behaviors.(16,17,20) Not until recently did the pharmaceutical industry realize the importance of packaging medications to easily facilitate rapid identification of and discrimination between potent drugs used in operating rooms. For years muscle relaxants such as pancuronium vials were very similar to those of heparin. Some manufacturers continue to package ephedrine in ampoules similar to those of oxytocin and epinephrine. This problem also occurs with different doses of the same drug--the vials for at least three concentrations of atropine sulfate from one manufacturer are similar. This results in inadvertent over- and under-dosing.
Any medication drawn into a syringe for later use should be labeled immediately. Unlabeled and incorrectly labeled syringes invite errors in drug administration and dosing and should be discarded. Routine use of approved, commercial color-coded labels may reduce these errors. The labels should conform to the standards of the American Society for Testing and Materials (ASTM).(18)
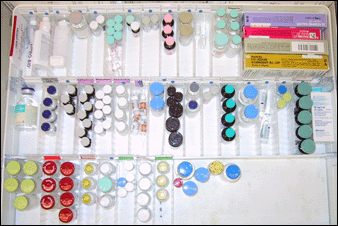
A cluttered and disorganized workspace also predisposes to medication errors and searches that can delay administration of emergency medications. All anesthesia and resuscitation medication carts should be standardized,(Figure 2) by applying a systematic method for stocking new and discarding outdated medications.
Case & Commentary: Part 3
Hand ventilation was continued to achieve normocapnia until the muscle relaxant had dissipated and neostigmine could be administered. After reversal of muscle relaxation, apnea resolved, anesthesia was discontinued, and the patient was transported safely to the post-operative care unit, where he recovered fully and was discharged.
To understand the causes of errors, we must examine what happened, what was the root cause, and what were the underlying system failures.(19) In a systems analysis, people are viewed as an important safety resource, not only a source of errors. Designing robust transparent systems, with built in feedback control strategies is important given human flexibility and fallibility. This was a case of unintentional administration of a paralytic agent in place of an antibiotic due to similar packaging. System checks that could be implemented here to avoid inadvertent drug swaps include color-coded labeling and reorganization of the anesthesia cart.(Table 4) Training all healthcare professions in the Six Right’s—patient, drug, dose, route, time, concentration—is critical to effective and safe medication administration. Recognizing environmental factors that predispose and distract clinicians is paramount. These include: noise, interruptions, fatigue and lack of adequate rest, poor lighting, and poor information systems.
Barcoding point-of-care systems (BPOC) are the main technology interventions to prevent medical and blood transfusion errors. The Institute of Medicine report noted that barcoding “is an effective remedy” for medication errors, “a simple way to ensure that the identity and dose of the drug are as prescribed, that it is being given to the right patient, and that all of the steps in the dispensing and administration processes are checked for timeliness and accuracy.” BPOC uses bedside computers that interact with a radio wave-controlled wireless communication system. Changes in medications and other patient information are instantly communicated from hospital information systems to the bedside unit, notifying nurses of changes. BPOC systems enable managers to monitor the medications given to patients and help hospitals identify opportunities for improvements in their medication-administration procedures.
Whenever medications are administered, robust identification systems must be present to avoid inadvertent wrong drug administration.
Take-Home Points
- Medication errors are the number one cause of preventable adverse events, including death.
- Causes of wrong drug administration include failure to label medications, mislabeling of syringe or ampoules, or failure to confirm identification of the medication by reading label carefully.
- To reduce drug administration errors in the OR, label syringes carefully with color-coded, pre-printed labels that conform to ASTM standards; use “ready-to-use” easily identified syringes to administer emergency drugs; standardize location of medications on the anesthesia cart; and always review the Six Right’s (patient, drug, dose, route, time, concentration).
- System checks should be designed into the medication administration process to prevent or reduce chances of inadvertent drug/vial swap.
- Physician drug order entry and barcoding systems can reduce the number of adverse drug events.
Paul Barach, MD, MPH University of Chicago
Faculty Disclosure: Dr. Barach has declared that neither he, nor any immediate member of his family, has a financial arrangement or other relationship with the manufacturers of any commercial products discussed in this continuing medical education activity. In addition, his commentary does not include information regarding investigational or off-label use of pharmaceutical products or medical devices.
References
1. Gravenstein Nick, ed. Manual Of Complications During Anesthesia. Lippincott, Philadelphia; 1991.
2. Cooper JB. Toward prevention of anesthetic mishaps. Int Anesthesiology Clin. 1984;22:167-183.[ go to PubMed ]
3. Lunn JN, Devlin HB. Lessons from the confidential enquiry into perioperative deaths in three NHS regions. Lancet. 1987;12:1384-1386.[ go to PubMed ]
4. Kohn LT, Corrigan JM Donaldson MS, eds. To Err is Human: Building a Safer Health System. Institute of Medicine. Committee on Quality of Health Care in America. Washington, DC: National Academy Press; 1999.
5. Bates DW, Cullen D, Laird N, et al. Incidence of adverse drug events and potential adverse drug events: implications for prevention. JAMA. 1995;274:29-34.[ go to PubMed ]
6. Leape LL, Brennan TA, Laird N, et al. The nature of adverse events in hospitalized patients: results of the Harvard Medical Practice Study II. N Engl J Med. 1991;324:377-384.[ go to PubMed ]
7. National Association of Insurance Commissioners. Medical Malpractice Closed Claims, 1975-1978. Brookfield, Wis; National Association of Insurance Commissioners; 1980.
8. Leape LL, Bates DW, Cullen DJ, Cooper J, Demonaco HJ, Gallivan T, et al. Systems analysis of adverse drug events. JAMA. 1995;274:35-43.[ go to PubMed ]
9. Reason JT, Carthey J, and de Leval, MR. Diagnosing “Vulnerable System Syndrome”: An Essential Prerequisite to Effective Risk Management. Qual Health Care. 2001;10:ii21-ii25.[ go to PubMed ]
10. Currie M, Mackay P, Morgan C, et al. The “wrong drug” problem in anesthesia: an analysis of 2000 incident reports. Anaesth Intensive Care. 1993;21:596-601.[ go to PubMed ]
11. Russell WJ. Getting in the red: a strategic step for safety. Qual Saf Health Care. 2002;11:107.[ go to PubMed ]
12. Cooper JB, Newbower RS, Kitz RJ. An analysis of major errors and equipment failures in anesthesia management: Considerations for prevention and detection. Anesthesiology. 1984;60:34-42.[ go to PubMed ]
13. Tissot E. Cornettte C, Demoly P. et al. Medication errors at the administration stage in an intensive care unit. Intensive Care Med. 1999;25:353-359.[ go to PubMed ]
14. Gaba DM, Maxwell M, DeAnda A. Anesthetic mishaps: breaking the chain of accident evolution. Anesthesiology. 1987;66:670-676.[ go to PubMed ]
15. Smith JW, Seidl LG, Cluff LE. Studies on epidemiology of adverse drug reactions. V. Clinical factors influencing susceptibility. Ann Intern Med. 1966;65:629-640.[ go to PubMed ]
16. Reason J. A preliminary classification of mistakes. In: Rasumussen J, Duncan K, Lelpat J, eds. New Technology And Human Error. Chiseter, Wiley;1987:15-22.
17. Rasmussen J. Information processing and human-machine interaction: an approach to cognitive engineering. New York: Elsevier;1986:149-151.
18. American Society for Testing and Materials. Standard specification for user applied drug labels in anesthesiology. American Society for Testing and Materials. D477494,1995.
19. Gaba DM, Fish K, Howard S, eds. Crisis Management in Anesthesiology. New York, NY: Churchill Livingston; 1994.
20. Bates, DW. Unexpected hypoglycemia in a critically ill patient. Ann Intern Med. 2002;137:110-116.[ go to PubMed ]
Tables
Table 1. When to Suspect Wrong Drug Administration in the Operating Room
| Unusual response or lack of response to drug administration: pounding heart, mental status changes, apnea, muscle weakness, and visual disturbances |
| Extreme or unexpected increase or decrease in blood pressure or heart rate |
| Unexpected or persistent muscle relaxation |
| Unexpected change, or lack of change, in level of consciousness |
| Incorrect ampoule found to be open in work area |
Table 2. Checklist: Steps to Determine Drug Administered
| Check the syringes and ampoules used during the case |
| Check to see if low volume unexpectedly remains in syringe |
| Inspect open ampoules |
| Impound the “Sharps” container to allow inspection of ampoules and syringes at later time |
| Consider drawing blood levels to ascertain drug given |
Table 3. Clinical Management of Apnea
|
Table 4. System Checks to Prevent Wrong Drug Administration
| Check for correct patient, drug name, concentration, dose, route, time |
| Use drug labels that conform to ASTM standards |
| Label syringes carefully—use preprinted color-coded adhesive labels |
| For emergency drugs, use “ready to use” syringes that are prepared according to ASTM standards |
| Standardize location of medications |
| Discard unlabeled vials, syringes |
Figures
Figure 1. Example of Similar-Looking Vials
Figure 2. View of Medication Cart Drawer