“This is the wrong patient's blood!”: Evaluating a Near-Miss Wrong Transfusion Event
Barnhard S. “This is the wrong patient's blood!”: Evaluating a Near-Miss Wrong Transfusion Event. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2020.
Barnhard S. “This is the wrong patient's blood!”: Evaluating a Near-Miss Wrong Transfusion Event. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2020.
The author(s) and reviewers for this Spotlight Case and Commentary have disclosed no relevant financial relationships with commercial interests related to this CME activity.
Case Objectives
- Identify the key aspects of the closed-loop blood delivery pathway and how they ensure transfusion recipient safety.
- Differentiate the human and technologic roles involved in delivering the correct blood to the correct patient.
- Recognize the potential system areas of risk.
- Identify areas to focus on for continuous quality improvement to ensure safe transfusion practices.
The Case
A 74-year-old male with a history of hypertension, hyperlipidemia, paroxysmal atrial fibrillation, coronary artery disease, congestive heart failure with an ejection fraction of 45%, stage I chronic kidney disease and gout presented for a total hip replacement. His home medications included lisinopril, metoprolol, colchicine, sertraline, acetaminophen and oxycodone as needed, and warfarin, which was withheld appropriately prior to the surgery.
The patient was seen by the surgical and anesthesia teams in the preoperative holding area the morning of surgery. An intravenous (IV) line was placed and a “type and cross for blood” request was sent along with baseline laboratory tests. At our institution, an initial blood sample is sent in a purple tube from the holding area and then the blood bank will request a second confirmatory sample in a pink tube. The anesthesiologist marks the first tube with a patient sticker, date, time, and initials. The blood bank then sends a pink tube with pre-made labels to the operating room (OR) for a second blood sample.
Shortly into the case, the patient became hypotensive and vasopressors were initiated. During this time, the patient's pink tube for the confirmatory blood sample was delivered to the room. The anesthesiologist filled the tube with blood and sent it back to the blood bank. About an hour into the case significant bleeding was encountered and a blood transfusion was needed. The patient information on the blood bags was checked per institution policy, which requires a witness signature. It was quickly discovered that the blood delivered contained the wrong labels. The blood bank was notified, the blood returned, and a new blood sample sent. Because the patient was persistently hypotensive and still bleeding, a massive transfusion protocol was initiated to rapidly get blood to the room. Uncrossed universal donor blood was delivered and administered, and the patient's hemodynamic parameters recovered appropriately.
The Commentary
By Sarah Barnhard, MD
Introduction: Transfusion Safety
The history of the blood supply in the U.S. is one of early, sobering frequency of disease transmission but also remarkable improvement in safety in terms of infectious agents.1 Rates of transfusion-transmitted viral infections such as the human immunodeficiency virus (HIV) and hepatitides have plummeted over the last few decades mainly due to the highly sensitive technique of viral nucleic acid testing. According to the National Blood Collection and Utilization Survey (NBCUS), 11,349,000 (95% confidence interval, 10,592,000–11,747,000) red blood cell units were transfused in the US in calendar year 2015.2 A case of transfusion-transmitted HIV has not been reported to the Centers for Disease Control and Prevention (CDC) since one traced to a 2008 donation3 and this single case was the first since 2002. The estimated risk of HIV transmission due to blood transfusion is 1 in 1.5 million (based on incidence estimate data from a selected time period during 2007-2008).4 This risk estimate is almost certainly too high because over 100 million red blood cell units have been transfused since 2008 without any documented cases of HIV transmission.
Still, in day-to-day counseling of patients regarding the risks of a red blood cell transfusion, most mention their apprehension due to the potential risk of HIV transmission. Some even recognize the risks of transfusion reactions (allergic, febrile non-hemolytic, etc.) defined by the National Healthcare Safety Network.6 Very few patients readily recognize systemic errors in the closed-loop blood delivery pathway as a serious risk to patient outcomes.
Transfusion System Infrastructure
The pathway of blood delivery is inherently complex because multiple patient care areas are involved. The goal of the blood delivery pathway is to deliver the right product to the right patient. The pathway can be summarized by three simple steps:
1) Identify the patient with two unique identifiers.
2) Connect the patient identifiers to all prepared lab samples, tests, and blood products.
3) Deliver the right blood product to the right patient at the right time, confirming patient ID again.
These three simple steps comprise numerous processes, each with its risk of failure. The highest rates of failure are associated with processes outside of the clinical laboratory.7
The clinical laboratory is one of the most highly regulated services in the hospital, and the transfusion service is one of the most highly regulated services within the clinical laboratory. The Centers for Medicare & Medicaid Services (CMS) regulates all laboratory testing (except research-related testing) performed on humans in the US through the Clinical Laboratory Improvement Amendments (CLIA).7 The Food and Drug Administration’s (FDA’s) Center for Biologics Evaluation and Research (CBER) regulates biological products for human use under applicable federal laws.8 The AABB (formerly American Association of Blood Banks), an international non-profit transfusion medicine organization, sets standards for transfusion medicine9and these are incorporated into state law in California10 and other states. The College of American Pathologists also sets standards for accreditation of clinical laboratories in compliance with CLIA.11 This robust oversight is aimed at ensuring quality laboratory testing and processes.
The Quality Management System
The American Society for Quality defines a quality management system (QMS) as “a formalized system that documents processes, procedures, and responsibilities for achieving quality policies and objectives.”12 For hospital-based transfusion services, a practical QMS is an essential part of meeting regulatory requirements and improving effectiveness and efficiency. AABB requires transfusion services to have documented QMSs (Standard 1.2).13
A hospital-based transfusion service QMS must include a closed-loop process that protects patients from an ABO-incompatible (blood type-incompatible) red blood cell transfusion. The loop begins and ends at the patient bedside. Notice how the following (paraphrased) AABB Standards protect a recipient from an ABO-incompatible red blood cell transfusion:13
- 5.11.1 All requests for blood contain two independent identifiers of the intended recipient.
- 5.11.2 All patient blood sample labels include two independent identifiers and (5.11.2.1) the label is affixed to the container before the person who obtained the sample leaves the bedside.
- 5.12 The ABO group of each donor unit of red blood cells is confirmed through serologic testing before being placed in stock inventory.
- 5.14.1 The ABO group of the patient is determined by comparing the ABO antigens detected with the presence of expected anti-A and anti-B antibodies.
- 5.16.1 Before issue, a crossmatch demonstrates ABO compatibility.
- 5.16.2 If a computer crossmatch technique is used, two determinations of the recipient’s ABO group must be made before transfusing non-group O red blood cell units.
- 5.14.5 The recipient’s historical records for ABO group are reviewed before every unit issued.
- 5.23 At the time a unit is issued, two people verify the recipient ABO group and the donor ABO group.
- 5.28.3 After issue and immediately before transfusion, two people verify the ABO group of the recipient and the donor ABO group and confirm recipient identification in the presence of the recipient. One of these two staff members must be the person transfusing the blood.
- 5.14.1 If a discrepancy is identified in the ABO testing, only group O red blood cells are transfused until resolution.
Risk of ABO-Incompatible Transfusions and Hemolytic Reactions
In the United States, reporting of fatalities to accreditors and regulators is mandatory but reporting of near-miss or system errors is not. Therefore, risk assessment is difficult due to underreporting and the fact that there is no centralized public database for tracking errors.
Employees at all hospital-based transfusion services across the US are very aware of the “Fatalities Reported to FDA Following Blood Collection and Transfusion Annual Summary”.14 All blood transfusion- and donation-related deaths must be reported to the FDA as soon as possible after confirming a complication of blood transfusion or donation. Such notifications must be followed by an investigation report within 7 days. Fatal acute hemolytic transfusion reactions related to ABO-mismatched transfusions were reported 1-4 times each year from 2013 to 2017.14
When combined with the NBCUS data, the risk of fatality due to an ABO-mismatched red blood cell transfusion could therefore be estimated at 1-4 per 10,000,000 for each red blood cell unit transfused. But fatal reactions represent the ‘tip of the iceberg’ as most ABO-incompatible transfusions involve small volumes due to early clinical signs/symptoms and usually patients survive.15
The risk of a lethal hemolytic transfusion reaction was estimated at 1 per 550,000 units transfused for the time period 1976-1985 in the US.16 Not all hemolytic reactions are ABO-related and not all wrong transfusion events result in adverse clinical outcomes. Others have estimated that 1 in every 19,000 units of red blood cells is transfused to the wrong patient each year, 1 in 76,000 transfusions results in an acute hemolytic reaction, and 1 in 1.8 million units of transfused red blood cell units results in death due to acute hemolytic reaction.17,18
When estimating risk, the best information available indicates that most transfusions to the wrong patient occur as a result of potentially avoidable system failures.19 The most frequent error leading to transfusion of ABO-incompatible blood occurs during patient identification/verification at the bedside; as a result, although the blood is labeled appropriately, it is transfused to someone other than the correct recipient.15
Response to a Near-Miss High-Risk Transfusion Event
For every process in the transfusion services laboratory, control measures should be in place to ensure quality outcomes. When process variation occurs, it is important to review the methods of process control and the need for improvement. “Process control” encompasses the defined activities that ensure a process is predictable, stable and consistently operating at a target performance level. Common cause variation is inherent in a process over time and requires constant process improvement, while special cause variation arises due to unusual circumstances and requires removing the cause.12
Per FDA, AABB and the College of American Pathologists (CAP), the response to a near-miss high-risk patient safety event in transfusion services must include:
- Notification of the appropriate accreditation and/or regulatory agencies if required; errors classified as blood product deviations (BPDs) must be reported within 45 calendar days to the FDA.20
- A broad root cause analysis:
- Evaluate standard operating procedures to determine if revision is needed.
- Interview staff involved to determine what aspects of the system failed and why.
- A documented corrective and preventative action plan (CAPA) submitted to laboratory leadership:
- Notify appropriate accreditation and/or regulatory agencies of CAPA if required.
- Retain the document for future inspections.
Root Cause Analysis of this Case
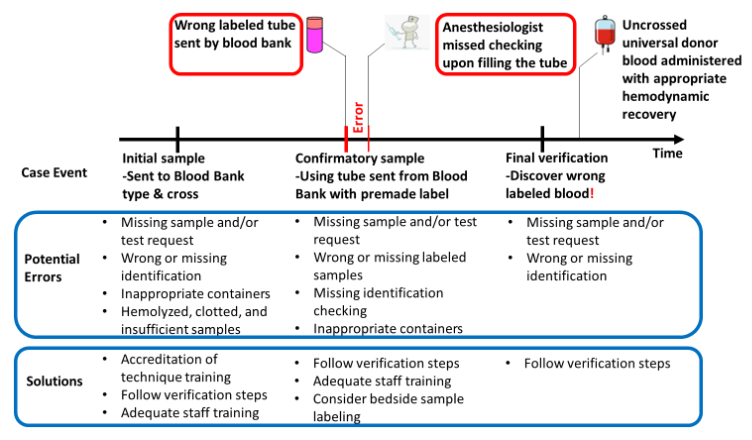
The root cause analysis of this Case (Figure 1) reveals two steps at which errors occurred: the tube was labeled incorrectly by the blood bank and was not checked at the bedside by the anesthesiologist before obtaining a blood sample. The underlying reasons for the two errors are not provided. The downstream effect of the two errors was the wrong blood reaching the patient’s bedside, a high risk near-miss event that may have led to serious patient outcomes.
Figure 1: Root cause analysis of this near-miss Case
Corrective and Preventative Action Plan (CAPA)
The underlying reasons for the two errors are not provided for this Case, but the general approach to the corrective and preventative action plan (CAPA) should include reviewing the standard operating procedures (SOP) with the involved staff to discuss whether there are any areas where the SOP is confusing or misleading. This review may include re-educating the staff on the current SOP, evaluating other samples processed by the involved staff members to ensure no other labeling errors were made, reviewing records for other deviations, assessing processes through direct observation of staff, or assessing skills with quizzes or other methods.
Re-addressing the six core elements of competency of the staff members involved, per CLIA, is required in the transfusion services laboratory when significant deviations occur. The core competency elements are: (1) direct observation of routine patient test performance; (2) monitoring the recording and reporting of test results; (3) review of intermediate test results, quality control records, proficiency testing results, and preventive maintenance records; (4) direct observation of performance of instrument maintenance and function checks; (5) assessment of test performance through testing of previously analyzed specimens, internal blind testing of samples, or external proficiency testing of samples; and (6) assessment of problem-solving skills.21 A monitoring process, typically through periodic audits, should be initiated if the underlying reason for the error is systemic and especially if a process change is made to ensure compliance.
One potential corrective action is to implement bedside sample labeling for all blood-type verifications to remove the variability of labels being generated from the laboratory. Checking the labels of all blood samples against the two unique identifiers on the patient’s wristband and labeling at the bedside is standard practice. Combining this standard practice with barcode scanning significantly reduces laboratory testing errors.22 Even without bedside labeling, barcode scanning of the patient’s wristband by the anesthesiologist at the time of sample collection should ensure the wristband and sample label match.
Approach to Urgent Transfusion Cases with Discrepancies
This Case highlights a realistic scenario in which clinical need was urgent despite system errors. It illustrates how sometimes patient care cannot await resolutions of discrepancies in the blood delivery pathway and when critical transfusions are required, emergent un-crossmatched red blood cells that are group O should be provided. Unfortunately, in this case, a massive transfusion protocol had to be initiated and un-crossmatched group O red blood cells provided to the patient in this Case, potentially causing a ‘universal blood’ shortage for a future patient. Although un-crossmatched group O units are the safest product to transfuse in emergent cases such as an unstable patient who is actively bleeding or who has significant organ dysfunction due to anemia, using un-crossmatched red blood cells increases the risk of hemolytic reaction due to non-ABO antibodies and should be avoided unless the clinical situation is truly emergent. Implementing strong systems processes as discussed in this Commentary should reduce the risk of errors in the transfusion delivery system and ensure that patients receive the correct blood product, reducing the need for un-crossmatched group O blood.
Take-Home Points
- Risk of error in the blood delivery pathway is significantly higher than the risk of transfusion-transmitted HIV or hepatitis in the U.S.
- Each step in the closed-loop blood delivery pathway is critical for transfusion safety; the highest area of risk is bedside patient identification.
- No matter how urgent, the steps in the closed-loop blood delivery pathway must always be followed to protect a patient from a fatal ABO-mismatched transfusion.
- In critically ill patients who require transfusion and cannot wait for verified, crossmatched blood to be available, only group O red blood cells should be transfused.
- Transfusion services are highly regulated services in the clinical laboratory that are under state and federal oversight.
- The appropriate response to a near-miss high-risk transfusion event includes: (1) report the event to accreditation/regulatory agencies as required, (2) perform a root cause analysis, (3) develop and implement a corrective and preventative action plan and (4) monitor the system.
Sarah Barnhard, MD
Medical Director of Transfusion Services
Department of Pathology and Laboratory Medicine
UC Davis Health
Acknowledgements: The author acknowledges the contribution of Ying Liu, MD, and Nam Tran, PhD for their assistance on the Root Cause Analysis.
References:
- Dzik WH. Emily Cooley Lecture 2002: Transfusion safety in the hospital. Transfusion. 2003; 43:1190-1199. [Available at]
- Ellingson KD, Sapiano MRP, Haass KA, et al. Continued decline in blood collection and transfusion in the United States-2015.Transfusion. 2017 Jun; 57(Suppl 2):1588-1598. [Free full text]
- Centers for Disease Control and Prevention (CDC). HIV transmission through transfusion --- Missouri and Colorado, 2008.MMWR Morb Mortal Wkly Rep. 2010;59(41):1335-1339. [Free full text]
- Zou S, Dorsey KA, Notari EP, et al. Prevalence, incidence, and residual risk of human immunodeficiency virus and hepatitis C virus infections among United States blood donors since the introduction of nucleic acid testing. Transfusion 2010; 50:1495-504. [Available at]
- Védy D, Robert D, Gasparini D,et al. Bacterial contamination of platelet concentrates: pathogen detection and inactivation methods. Hematol Rev 2009 Mar 1; 1(1):e5. [Free full text]
- Centers for Disease Control and Prevention (CDC). National Healthcare Safety Network Biovigilance Component Hemovigilance Module Surveillance Protocol. Accessed December 26, 2019. [Free full text (PDF)]
- Centers for Medicare and Medicaid Services (CMS). Clinical laboratory improvement amendments (CLIA). [Free full text]
- U.S. Food and Drug Administration (FDA). Center for Biologics Evaluation and Research (CBER). Accessed December 26, 2019. [Free full text]
- AABB. Transfusion Medicine.Accessed December 26, 2019. [Free full text]
- California Department of Public Health (CDPH). Blood banks and biologics.Accessed December 26, 2019.[Free full text]
- College of American Pathologists. Laboratory accreditation program. Accessed December 26, 2019. [Free full text]
- American Society for Quality (ASQ). What is a quality management system (QMS)? Accessed December 26, 2019. [Free full text]
- Standards for Blood Banks and Transfusion Services. AABB 31st Edition. Effective April 1, 2018. [Available at]
- Fatalities Reported to FDA Following Blood Collection and Transfusion. Annual Summary for Fiscal Year 2017. Accessed December 26, 2019. [Free full text]
- Janatpour KA, Kalmin ND, Jensen HM, et al. Clinical outcomes of ABO-incompatible RBC transfusions. Am J Clin Pathol. 2008; 129:276-281. [Free full text]
- Strobel E. Hemolytic transfusion reactions. Transfus Med Hemother. 2008; 35(5):346-353. [Free full text]
- Vamvakas EC, et al. Transfusion-related mortality: the ongoing risks of allogeneic blood transfusion and the available strategies for their prevention. Blood 2009; 113:3406-17. [Free full text]
- Fung MK, Grossman BJ, Hillyer CD and Westhoff CM, Eds. AABB Technical Manual 18th Edition 2014. Accessed January 6, 2020. [Free full text]
- Stainsby D, Jones H, Asher D, et al.Serious hazards of transfusion: a decade of hemovigilance in the UK. Transfus Med Rev. 2006; 20:273-282. [Free full text]
- Snyder SR, Favoretto AM, Derzon JH, et al.Effectiveness of barcoding for reducing patient specimen and laboratory testing identification errors: alaboratory medicine best practices systematic review and meta-analysis. Clin Biochem. 2012;45(13-14):988-998. [Free full text]
- FDA Blood Product Deviation Reporting for Blood and Plasma Establishments: Guidance for Industry. 2006 Oct. Accessed January 6, 2020. [Free full text]
- Medicare, Medicaid and CLIA programs; regulations implementing the Clinical Laboratory Improvement Amendments of 1988 (CLIA)--HCFA. Final rule with comment period. Fed Regist. 1992 Feb 28; 57(40):7002-186. [Free full text]