False Assumptions Result in a Missed Pneumothorax after Bronchoscopy with Transbronchial Biopsy.
Kuhn BT, Chau-Etchepare F. False Assumptions Result in a Missed Pneumothorax after Bronchoscopy with Transbronchial Biopsy.. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2022.
Kuhn BT, Chau-Etchepare F. False Assumptions Result in a Missed Pneumothorax after Bronchoscopy with Transbronchial Biopsy.. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2022.
Brooks T Kuhn, MD, disclosed a relevant financial disclosure with an ineligible company related to this CME activity which has been mitigated through UC Davis Health, Office of Continuing Medical Education procedures to meet ACCME standards.
| NAME | COMPANY | RELATIONSHIP |
|---|---|---|
| Brooks T. Kuhn | Takeda, Grifols Boehringer-Ingelheim, Inhibrx Inhibrx, Theravance |
Speaker Consultant Author |
Patrick Romano, MD, MPH; Debra Bakerjian, PhD, APRN, RN; Ulfat Shaikh, MD, Shelley Palumbo, MS, CCC-SLP, and Florence Chau-Etchepare, MD for this Spotlight Case and Commentary have disclosed no relevant financial relationships with ineligible companies related to this CME activity.
Learning Objectives
At the conclusion of this educational activity, participants should be able to:
- To identify complications associated with flexible bronchoscopy and transbronchial biopsy, especially iatrogenic pneumothorax
- To understand the role of checklists in assuring completion of routine safety-related clinical procedures
- To describe alternate methods of assessing patients for post-procedural pneumothorax without routine chest radiography
- To understand that the symptoms of pneumothorax, pain and dyspnea, are under-identified, under-quantified, and under-treated in Black patients due to implicit bias
The Case
A 47-year-old man underwent a navigational bronchoscopy with transbronchial biopsy for evaluation of lymphadenopathy, cough, and weight loss. The procedure was performed under general anesthesia, by an attending physician and a fellow, without complications. The patient was transferred to the post-acute care unit (PACU) for observation and a routine post-procedure chest x-ray (CXR). The patient recovered without change in respiratory symptoms or vital signs in the PACU.
After the CXR was taken, the attending physician spoke to the patient and discussed his impressions, although he had not yet seen the CXR. The attending physician left the PACU without communicating with the bedside nurse, who was caring for other patients. The patient informed the nurse that the attending physician had no concerns. While preparing the patient for discharge, the nurse paged the fellow requesting discharge orders. The fellow assumed that the attending physician had reviewed the CXR and submitted the discharge orders as requested. The patient was discharged home with family members, with instructions to follow up “as needed,” as his vital signs and respiratory symptoms remained normal.
Thirty minutes after discharge, the radiologist called the care team to alert them to the finding of pneumothorax on the post-procedure CXR. The patient was contacted to return to the PACU immediately. A follow-up CXR was performed and showed an enlarging pneumothorax. The patient was admitted overnight for observation and discharged the next day after treatment with supplemental oxygen; thoracostomy drainage was not necessary. After subsequent discussion among the care team members, it was apparent the fellow and attending each thought the other had reviewed the CXR. The bedside nurse presumed the CXR was read as normal after hearing from the patient that the attending had no concerns and thus proceeded with discharge. If not for the radiologist’s rapid communication to the care team, preventable harm from the pneumothorax could have occurred.
The Commentary
By Brooks T Kuhn, MD, and Florence Chau-Etchepare, MD
Post-Bronchoscopy Complications and Pneumothorax
Since its introduction over 50 years ago, flexible bronchoscopy has proven to be an effective and less invasive option for both diagnostic and therapeutic purposes in pulmonary medicine. Current-day bronchoscopy not only provides visualization of the lower respiratory tract but can also allow for various sampling techniques including washes or lavages, endobronchial biopsies for airway irregularities, transbronchial biopsies for nodules or parenchymal processes, and transbronchial needle aspirations to sample lymph nodes or other lesions. While bronchoscopy is very safe with low risk of mortality or serious adverse events, it does have associated complications of which clinicians should be aware.
Medications provided to induce various levels of sedation (from light to general anesthesia) can have hemodynamic consequences including hypotension and arrythmias in the peri-procedure period. Post-procedural coughing is a common complication with reported incidences up to 55%, although this symptom usually resolves within several days.1 Post-bronchoscopy elevation in body temperature occurs commonly (up to 33% of cases), but typically resolves without intervention and post-procedural infection is uncommon (6.5 to 7%).1,2 While potentially catastrophic, severe bleeding is quite rare. Carr et al. reviewed a cohort of 234 patients finding 89.7% of these patients had minimal bleeding (<5ml), 8.1% had mild (5-20 ml) blood loss, and 2.1% had moderate (20-100 ml) of blood loss.3
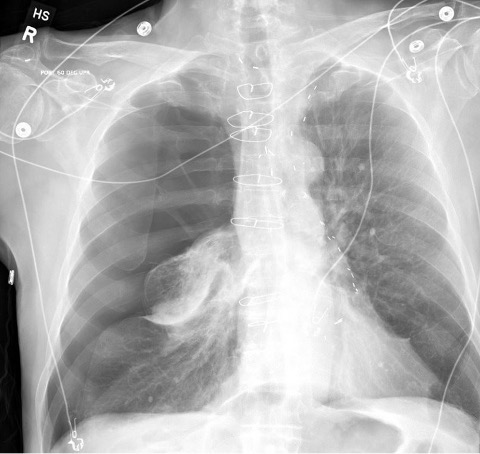
Iatrogenic pneumothorax is a known and feared complication of flexible bronchoscopy (Figure 1), with a reported incidence rate ranging from 0.16% when bronchoscopy alone is performed and up to 20-30% after placement of endobronchial valves for lung volume reduction. Whentransbronchial biopsy is performed—which is often the reason for using navigational bronchoscopy, as in this case—pneumothorax occurs at a higher incidence between 1% and 4%.4 The effects of pneumothorax vary widely, from an asymptomatic finding requiring only monitoring to “tension” pneumothorax with acute respiratory failure, shock, and even death if not treated with tube thoracostomy.5 Most commonly, patients complain of ipsilateral pleuritic chest pain and dyspnea. While no study directly assessing pneumothorax and bronchoscopy has been performed, pain and dyspnea have been shown to be under-identified, under-quantified, and under-treated in Black patients due to implicit bias.6
Figure 1. Large right pneumothorax after bronchoscopy
Risk factors for pneumothorax include the presence of emphysema and biopsy of non-solid targets and lung segments with a sagittal orientation, potentially due to more challenging visualization and more dynamic lobe movement associated with these circumstances.4,5Provider experience has not been shown to correlate with risk of pneumothorax from bronchoscopic procedures. Even with newer navigational technologies such as endobronchoscopic ultrasound (with and without sheaths) and robotic bronchoscopy, pneumothorax rates remain around 3%, suggesting that target location and characteristics affect risk more than proceduralist skill.4 Two approaches that may reduce the risk of pneumothorax include minimizing sedation to allow patients to communicate pain if forceps grab innervated pleura and using visualization with fluoroscopy or other means to assess forcep location.1 In the majority of cases, pneumothorax is treated with tube thoracostomy, but observation and conservative therapy is also an option for patients who are clinically stable.5
Improving Patient Safety and Communication Post-Transbronchial Biopsy
Though the use of an order set did not prevent this near-miss event, this case should not be perceived as an argument against the utility of checklists or order sets. Rather, it demonstrates a common scenario in which a busy medical team hands off or turns over care to another team member who may or may not understand what has already been done, the expected recovery trajectory, and the milestones that must be achieved before the patient can be safely discharged. In the increasingly complicated and fast-paced healthcare environment, clear and efficient communication is essential. Checklists and order sets, when designed and used appropriately, can help standardize and organize the most crucial aspects of care during these transitions. Checklists should provide answers to the basic questions of “who, what, where, when and how.” They can prompt team members to remind each other of delegated tasks and action items to ensure follow-through and task completion. Stress, fatigue, and the complexity of various procedures can adversely affect our memories, and multi-tasking can further reduce accuracy.6 The use of checklists can reduce the deleterious effects of juggling simultaneous demands. Checklists have been shown to be effective in multiple different clinical settings, for tasks such as identifying correct surgical sites in the operating room and reducing infections associated with central lines placed in hospitalized patients.7 However, the successful use of checklists depends on the continued, timely input of clinicians and care team members who use them to ensure that they maintain relevance and reflect current best practices.
The American College of Radiology (ACR) defines the presence of pneumothorax as a category 1 actionable finding and recommends reporting of those findings, within minutes and via verbal communication, directly to the appropriate clinicians.8 Despite these guidelines, significant time often lapses between images being obtained and findings being communicated to the ordering provider, due to image processing time, delays in uploading and reading the images, and delays in communication. In this case, potential harm was averted by the radiologist’s recognition and reporting of the pneumothorax, but the overall timing of these actions was not ideal. Given the immediate risk posed by a pneumothorax, even the rapid read and response from the radiologist was too late to ensure patient safety as it occurred after the patient was discharged.
The sign-out process typically involves a transfer of care to a new clinician or team member. In many situations, as in this case, the accepting team member is also caring for other patients who may also be “new” to them. The organized structure of a checklist, in conjunction with a verbally communicated handoff, can help assure that each team member understands their roles and responsibilities during the post-procedure recovery period and that specific clinical criteria have been met before discharge.
The necessity of a routine chest radiograph (CXR) after transbronchial biopsy is currently being questioned, although it is still a common practice among pulmonologists. Routine post-bronchoscopy CXRs rarely provide actionable information for patients without acute symptoms and expose patients to potentially unnecessary radiation.9 Alternatives to routine CXR after transbronchial biopsy do exist. For example, the British Thoracic Society recommends monitoring patients for two hours post-procedure, with CXR performed only if clinically warranted.9 However, selective imaging may increase disparities in care because of implicit bias in providers’ assessment of symptoms such dyspnea and chest pain. If imaging is performed based on signs and symptoms, “peer network support” and other educational and reflective interventions may be needed to mitigate the possible effects of implicit bias on how post-procedural symptoms are evaluated and interpreted.10 Recently, the use of ultrasound of the pleural interface has been shown to have higher sensitivity and specificity for the diagnosis of pneumothorax than CXR, specifically among patients after transbronchial biopsy as well as in other cohorts.11 Consequently, many clinicians are replacing routine CXR with post-procedural ultrasound to rapidly assess patients for pneumothorax at the bedside.
Iatrogenic pneumothorax is a known complication of flexible bronchoscopy, especially with transbronchial biopsy, and diagnostic vigilance after the procedure is therefore required to ensure patient safety. In this case, despite timely initiation of a process to screen for post-procedural pneumothorax with CXR, interpretation and communication of the CXR results did not happen until after the patient had been discharged. Various unavoidable circumstances, such as transfers of patients between procedure suites and post-care units, subsequent handoffs between nurses and staff, and the split attention of proceduralists, necessitate the use of standardized processes to assure that diagnostic tests are not just performed but also interpreted and documented in patients’ medical records. In this case, poor communication and confusion of responsibility resulted in a near-miss due to an expanding pneumothorax that required clinical attention. The quick turnover and potentially rapid expansion of pneumothorax requires post-bronchoscopy care teams to develop reliable systems for rapid assessment of pneumothorax at the point-of-care.
Take-Home Points
- Bronchoscopy is a safe, common procedure but, as with all procedures, complications such as hypotension, cough, bleeding, fever, and pneumothorax can arise.
- Iatrogenic pneumothorax is a relatively common complication after bronchoscopy, most often associated with transbronchial biopsies.
- Checklists and clear communication among team members — involving explanation of the procedure performed and any difficulties or complications encountered, criteria for safe discharge, and clear delegation of responsibilities — is vital to assure the safety of patients during and after bronchoscopy.
- If routine CXRs are taken after transbronchial biopsies for the evaluation of pneumothorax, they must be reviewed and their findings must be conveyed to the rest of the care team in a timely manner.
- Point-of-care ultrasound and/or symptom/vital sign-driven imaging are reasonable, and perhaps preferred, alternatives to CXR for assessing patients for pneumothorax at the point of care, including after transbronchial biopsy. However, selective imaging strategies may lead to missed pneumothoraxes in certain patient populations due to implicit bias.
Brooks Thomas Kuhn, MD, MAS
Assistant Professor of Clinical Medicine
Division of Pulmonary and Critical Care Medicine
UC Davis Health
btkuhn@ucdavis.edu
Florence Chau-Etchepare, MD
Assistant Professor of Clinical Medicine
Division of Pulmonary and Critical Care Medicine
UC Davis Health
fvchau@ucdavis.edu
References
- Leiten EO, Martinsen EMH, Bakke PS, et al. Complications and discomfort of bronchoscopy: a systematic review. European Clin Respir J. 2016;3:33324. [Free full text]
- Krause A, Hohberg B, Heine F, et al. Cytokines derived from alveolar macrophages induce fever after bronchoscopy and bronchoalveolar lavage. Am J Resp Crit Care. 1997;155:1793–1797. [Available at]
- Carr IM, Koegelenberg CFN, von Groote-Bidlingmaier F. Blood loss during flexible bronchoscopy: aprospective observational study. Respiration. 2021;84:312–318. [Free full text]
- Gotoh Y, Yamaguchi T, Yatsuya H, et al. Predictive risk factors for pneumothorax after transbronchial biopsy using endobronchial ultrasonography with a guide sheath. BMC Pulm Med. 2021;21(1):181. [Free full text]
- Bensted K, McKenzie J, Havryk A, et al. Lung ultrasound after transbronchial biopsy for pneumothorax screening in post–lung transplant patients. J Bronchol Interventional Pulmonol. 2018;25:42–47. [Free full text]
- Hoffman KM, Trawalter S, Axt JR, et al. Racial bias in pain assessment and treatment recommendations, and false beliefs about biological differences between blacks and whites. Proc National Acad Sci. 2016;113:4296–4301. [Free full text]
- Winters BD, Gurses AP, Lehmann H, et al. Clinical review: Checklists - translating evidence into practice. Crit Care. 2009;13:210–210. [Free full text]
- Allen B, Chatfield M, Burleson J, et al. Improving diagnosis in health care: perspectives from the American College of Radiology. Diagnosis. 2017;4:113–124. [Free full text]
- Rand IAD, Blaikley J, Booton R, et al. British Thoracic Society guideline for diagnostic flexible bronchoscopy in adults: accredited by NICE. Thorax. 2013;68:i1. [Free full text]
- Centola D, Guilbeault D, Sarkar U, et al. The reduction of race and gender bias in clinical treatment recommendations using clinician peer networks in an experimental setting. Nat Commun. 2021;12:6585. [Free full text]
- Kumar S, Agarwal R, Aggarwal AN, et al. Role of ultrasonography in the diagnosis and management of pneumothorax following transbronchial lung biopsy. J Bronchol Interventional Pulmonol. 2015;22:14–19. [Free full text]