Paradoxical Pulse
The Case
An 80-year-old man with paroxysmal atrial fibrillation and symptomatic bradycardia underwent successful pacemaker placement as an outpatient. The patient was restarted on his warfarin therapy and returned home with no complaints.
A week later, the patient developed new diffuse chest discomfort that progressed over the ensuing 24 hours to include associated dyspnea and nausea. He presented to the emergency department (ED) for evaluation where he was noted to be afebrile with a blood pressure of 113/66, heart rate of 95, and an oxygen saturation of 99% on room air. On physical examination, he appeared comfortable and in no distress. His cardiac exam was notable only for flat neck veins, an irregularly irregular rhythm, a well-healed pacer site, and no peripheral edema. Laboratory studies revealed a stable hemoglobin level, a negative troponin, and an INR of 2.6 (within the therapeutic range), but a slightly elevated creatinine and a new transaminitis (elevated liver tests) compared to a week ago. An electrocardiogram showed atrial fibrillation but no evidence of ischemia. Imaging studies included a chest x-ray, which was normal, and a computed tomography (CT) scan of the abdomen, which revealed a pericardial effusion but no hepatobiliary pathology.
The ED physician discussed the case and CT findings with the on-call cardiologist who recommended admission but no need for an urgent echocardiogram given the normal vital signs and the patient's clinical stability. The following day, the patient's echocardiogram confirmed a large pericardial effusion "without tamponade" physiology. Providers continued to remark about the patient's clinical stability based on vital signs and appearance. Later that day, the patient became acutely hypotensive, developed ventricular tachycardia and pulseless electrical activity, and required emergent resuscitative measures, including large drainage of a pericardial effusion to relieve his cardiac tamponade. While surviving the emergent resuscitation, the patient ultimately suffered additional complications and passed away after a prolonged hospital course.
The Commentary
Clinicians are often reassured by the general appearance of a patient, "stable" vital signs, and a benign physical exam. In some clinical scenarios, however, this heuristic fails: patients "look fine," and by the time the patient does not "look fine," it is too late. Thus knowing the limitations of the clinical examination is often important. In this case, an elderly gentleman on warfarin had a pacemaker implanted and subsequently developed cardiac tamponade. His large pericardial effusion, which was presumably the result of the "post-pericardiotomy syndrome" reported with transvenous procedures (1), was likely exacerbated by his anticoagulation therapy. The pericardial effusion was discovered, but the patient's clinical appearance, vital signs, and echocardiogram reassured his physicians. Unfortunately, his effusion progressed to cause hemodynamic compromise from which he ultimately succumbed.
The clinical examination of the cardiovascular system is challenging to trainees and experienced clinicians alike. Despite the countless hours spent by students, residents, and faculty gazing at neck veins, the overall accuracy of jugular venous pressure (JVP) in predicting low, normal, or high central venous pressure is only 56%.(2) The presence of an S3 in a patient older than 40 is widely accepted as a sign of left-sided heart failure, but this finding is plagued by high interobserver variability: the chances that an S3 heard by one cardiologist will be heard by another cardiologist is only 38%.(3) In contrast, the absence of the early diastolic murmur of aortic regurgitation is very helpful in ruling out moderate or severe aortic regurgitation (LR- 0.1), and the presence of this murmur is very helpful in ruling it in (LR+ 8.8), with excellent agreement among experienced examiners.(4) Yet despite its challenges and inaccuracies, we rely on the bedside clinical examination to detect urgent situations such as the one that developed in this case.
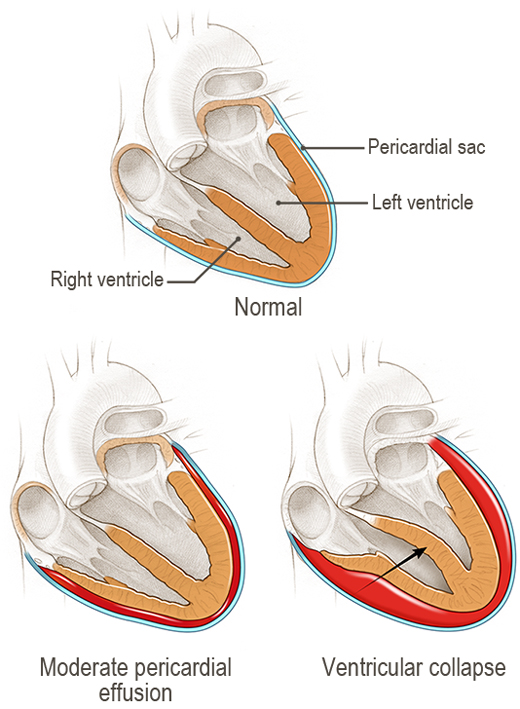
Cardiac tamponade occurs when fluid or gas trapped in the pericardial space compresses the heart and causes hemodynamic compromise (Figure). This syndrome exists on a continuum, from barely perceptible hemodynamic effects to overt collapse.(5-6) Beck's triad of hypotension, a quiet precordium, and increasing JVP was described in surgical patients who abruptly bled into their pericardium.(7) This triad is seldom seen in medical patients, who usually have a slow build up of pericardial fluid, which allows the pericardium to compensate by stretching.(8) As in this case, such patients may "look" stable even as fluid is accumulating in the sack that surrounds their heart.
The clinical examination of cardiac tamponade starts with a history. In this case the key points were recent pacemaker placement, anticoagulation therapy, and symptoms of chest discomfort, dyspnea, and nausea. The symptoms are fairly non-specific, but in the context of the recent procedure and anticoagulation, the clinicians should have had a high suspicion that the patient was at risk for a progressive pericardial effusion. However, when they discovered the pericardial effusion on a CT done for another reason, they were not willing to attribute his symptoms to this finding. Perhaps this was because he did not "look ill," or because their primary motivation to obtain the CT was to investigate his abnormal liver enzymes, not his heart. In this scenario, a small pericardial effusion might be considered an "incidentaloma," and perhaps might even have been considered to be "normal" after pacemaker placement. It was probably a combination of all these lines of reasoning, in addition to over-the-phone reassurance by the on-call cardiologist, that led them to not seriously consider tamponade in their differential diagnosis.
The clinicians were also falsely reassured by negative physical exam findings. They noted the absence of hypotension, but even in patients with known tamponade, the pooled sensitivity of this finding is only 26%, and indeed, some patients with tamponade have been known to be hypertensive.(9-10) The JVP was not thought to be elevated, but the accuracy and precision of this finding is not high, and the pooled sensitivity of elevated JVP in patients with known tamponade is only 76%.(9) The hallmark physical exam finding of cardiac tamponade is the pulsus paradoxus, which does not appear to have been performed in this case. Pulsus paradoxus is an exaggeration of the physiologic drop in blood pressure with inspiration. Originally described in 1873 by Adolf Kussmaul as a palpable diminution of the radial pulse with inspiration, this finding is typically elicited with a sphygmomanometer. The cuff is deflated slowly while listening for the first Korotkoff sounds, which are intermittent and disappear with inspiration. The cuff is then deflated further until the sounds become constant with each cardiac cycle. The difference between these two pressures is the pulsus paradoxus. A pulsus paradoxus is considered abnormal if it is greater than 10 mm Hg. In one study a pulsus paradoxus of greater than 10 mm Hg had a LR- of 0.03; hence, the absence of this sign makes cardiac tamponade unlikely.(11)
Echocardiography is the non-invasive test of choice for the diagnosis of cardiac tamponade; it has largely replaced cardiac catheterization and direct measurement of intracardiac pressures. Echocardiographic findings of right atrial and right ventricular collapse and inferior vena cava plethora are consistent with cardiac tamponade.(12) Certain findings on echocardiography actually confirm the phenomenon of pulsus paradoxus (exaggerated respirophasic changes in mitral valve and tricuspid valve inflow velocities). In this case, the initial echo was reassuring, as was the clinical appearance of the patient. However, pericardial fluid was likely continuing to accumulate as the patient bled into the closed pericardial space.
Knowing that this patient had a large pericardial effusion that was at risk of progressing, the clinicians should have been looking for early evidence of frank hemodynamic collapse by regular checks for pulsus paradoxus with a sphygmomanometer. They also might have followed for pulsus paradoxus by using an arterial line tracing, or even by using continuous pulse oximetry, which will pick up respirophasic changes in the pulse waveform.(13) Thus the clinicians in this case also fell prey to another heuristic of "blind obedience" to technology; a reassuring echocardiogram at a single point in time made them complacent and prevented them from following a simple bedside exam finding.
Take-Home Points
- It is as important to know the limits of the clinical examination as it is to know when it is helpful.
- Clinicians must be aware that the heuristic of, a patient who "looks fine" therefore must "be fine," will sometimes lead to incorrect assumptions.
- Cardiac tamponade in medical patients is a continuum of hemodynamic effects and can progress slowly due to compensatory mechanisms.
- While echocardiography is the non-invasive reference standard for the detection of cardiac tamponade, the pulsus paradoxus can be a helpful bedside test in ruling out tamponade or following patients with a known pericardial effusion.
Christopher Roy, MD Assistant Professor of Medicine Harvard Medical School
References
1. Miller GL, Coccio EB, Sharma SC. Postpericardiotomy syndrome and cardiac tamponade following transvenous pacemaker placement. Clin Cardiol. 1996;19:255-256. [go to PubMed]
2. Cook DJ, Simel DL. The Rational Clinical Examination. Does this patient have abnormal central venous pressure? JAMA. 1996;275:630-634. [go to PubMed]
3. Joshi N. The third heart sound. South Med J. 1999;92:756-761. [go to PubMed]
4. Choudhry NK, Etchells EE. The rational clinical examination. Does this patient have aortic regurgitation? JAMA. 1999;281:2231-2238. [go to PubMed]
5. Reddy PS, Curtiss EI, Uretsky BF. Spectrum of hemodynamic changes in cardiac tamponade. Am J Cardiol. 1990;66:1487-1491. [go to PubMed]
6. Fowler NO. The significance of echocardiographic-Doppler studies in cardiac tamponade. J Am Coll Cardiol. 1988;11:1031-1033. [go to PubMed]
7. Beck C. Two cardiac compression triads. JAMA. 1935:714-716. [Available at]
8. Fowler NO. Physiology of cardiac tamponade and pulsus paradoxus. I: mechanisms of pulsus paradoxus in cardiac tamponade. Mod Concepts Cardiovasc Dis. 1978;47:109-113. [go to PubMed]
9. Roy CL, Minor MA, Brookhart MA, Choudhry NK. Does this patient with a pericardial effusion have cardiac tamponade? JAMA. 2007;297:1810-1818. [go to PubMed]
10. Brown J, MacKinnon D, King A, Vanderbush E. Elevated arterial blood pressure in cardiac tamponade. N Engl J Med. 1992;327:463-466. [go to PubMed]
11. Curtiss EI, Reddy PS, Uretsky BF, Cecchetti AA. Pulsus paradoxus: definition and relation to the severity of cardiac tamponade. Am Heart J. 1988;115:391-398. [go to PubMed]
12. Singh S, Wann LS, Schuchard GH, et al. Right ventricular and right atrial collapse in patients with cardiac tamponade—a combined echocardiographic and hemodynamic study. Circulation. 1984;70:966-971. [go to PubMed]
13. Hartert TV, Wheeler AP, Sheller JR. Use of pulse oximetry to recognize severity of airflow obstruction in obstructive airway disease: correlation with pulsus paradoxus. Chest. 1999;115:475-481. [go to PubMed]
Figure
Figure. Normal cardiac anatomy with a progression from a moderate pericardial effusion to one with tamponade physiology due to ventricular collapse.
(Illustration © 2011 by Chris Gralapp.)