Who Nose Where the Airway Is?
The Case
A 70-year-old man with peripheral vascular disease was brought to the operating room to undergo vascular bypass surgery on his right upper extremity. Because the surgery was expected to involve only the arm, the case was started using local anesthesia. A certified registered nurse anesthetist (CRNA) was present to monitor the patient and to provide intravenous medications for anxiety, sedation, and pain control (monitored anesthesia care [MAC]). The CRNA was supervised by an attending anesthesiologist (a physician) but, because the surgery was a straightforward procedure performed under local anesthesia with MAC, the anesthesiologist was not physically present and knew very little about the case.
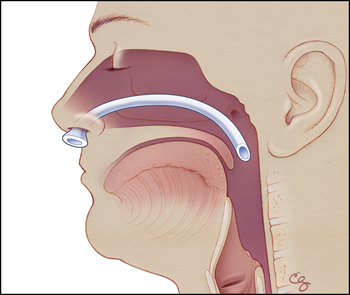
During the procedure, the patient complained of generalized discomfort and anxiety, requiring increased doses of opiates and benzodiazepines (an anti-anxiety medication). With these medications, the patient became sleepy but remained arousable. To maintain a patent airway, the CRNA placed a nasopharyngeal airway, a soft plastic tube inserted into the nose that extends into the posterior pharynx (Figures). Normally, the tip of this tube remains outside the nose, where it can be seen by clinicians. After this insertion, the patient did well with normal respirations and oxygen saturations.
After 2 hours of surgery, the vascular surgeon decided that they would need to use a vein from the patient's legs to replace a blood vessel in his upper extremity. Because this was expected to be a long and relatively complicated procedure, the decision was made to switch the patient to generalized anesthesia, which would require endotracheal intubation (placement of a plastic tube through the mouth and down into the lungs) and mechanical ventilation. The CRNA contacted the supervising anesthesiologist to help convert the case to general anesthesia. Very little information was exchanged between the two clinicians, and the anesthesiologist intubated the patient without complication. The CRNA managed the anesthesia, and the remainder of the surgery was uneventful. The patient was successfully extubated and did well after the surgery.
The following day, the patient remained stable, and the physician team was planning on discharging him to home. Curiously, the patient complained to the team that although he was feeling well, when he tried to drink any liquid it would come right out of his nose. To overcome the skepticism of the surgical team, the patient took a gulp of his orange juice with the physicians present, and, sure enough, most of the juice flowed out of his nose onto his hospital gown. To the astonishment of the team, he repeated the scenario.
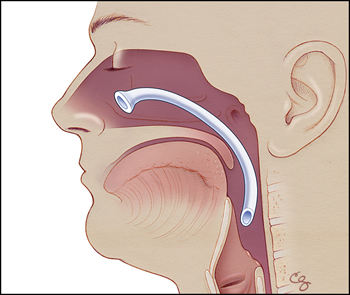
The surgeons decided to make the patient NPO (nothing by mouth) and gave him fluids intravenously. They consulted otolaryngology (ear, nose, and throat specialists) with concerns about a pharyngeal fistula or another anatomic abnormality—there were no obvious abnormalities seen extruding from the nose. The otolaryngologist discovered that the nasopharyngeal airway, which had been placed during the initial surgery, was still in his nose. It was not entirely clear how the tube had become lodged within the nasal cavity (so that its external portion was no longer extruding from the nose), but presumably this occurred when the patient was intubated by the anesthesiologist.
The plastic tube was removed, and the patient was discharged home later that day. He suffered no significant consequences from the event, but he did require an additional day in the hospital.
The Commentary
This case highlights a known hazard and complication of airway management: airway device aspiration. A number of different devices are employed to maintain patent airways during surgical procedures, but among the most common is a nasopharyngeal airway (NPA). In the course of perioperative airway management, it is possible for the nasopharyngeal airway to be aspirated or to become lodged in the airway (essentially becoming a retained foreign object [RFO]). Although the incidence of airway device aspiration is not known from any systematic study, both published case reports (1-4) and anecdotal evidence suggest that what happened in the presented case is not a unique occurrence. Health care providers involved in perioperative care and airway management should be aware of the risk factors for, clinical presentation of, and strategies to prevent airway device aspiration.
Specific risk factors for airway device (NPA) aspiration are not known from the literature, but coughing and sneezing may increase the risk because of the strong inspiratory force generated immediately preceding the cough or sneeze. Patients who have aspirated airway devices can have many different clinical presentations. Although the patient in this case presented rather comically with orange juice pouring from his nose, far more serious complications have been reported. Unsurprisingly, foreign bodies that completely or significantly obstruct the airway are more rapidly symptomatic. However, with aspirated airway devices, the presentation may be more insidious because the open lumen in the airway device may allow air to pass with little or no obvious immediate obstruction.(1-3) Providers should be aware that patients who have undergone procedures involving NPAs are at risk for immediate and delayed airway complications. In one case, the discovery of the aspirated NPA was delayed for weeks after the presumed event and was discovered only after persistent cough and recurrent chest infections triggered further evaluation. The device was lodged within the trachea and right bronchus.(1) One death was reported in a case with an aspirated airway, although this occurred in the context of significant underlying respiratory failure.(4)
As these cases illustrate, intubation and airway management are invasive procedures that carry some risk of an RFO. Strategies to prevent complications associated with these events include both device modifications and changes of practice to allow for early recognition of an airway aspiration or RFO event.
Device Considerations
Two basic NPA designs exist—one with a fixed flange at the distal end of the device, and the other with a movable flange to allow for more individualized fitting (Figures). It has been suggested that the adjustable flange may increase the risk of aspiration (2), but events have been reported with both designs. Multiple authors have suggested device modifications that might prevent aspiration of NPAs. Modifications include attachments to the external portion of the NPA that would rest outside of the patient's nose, which could make it more difficult for the device to be pulled or pushed completely into the patient. One group suggested the use of a safety pin (5), while another recommended the use of an endotracheal tube connector.(6) While possibly useful, the benefits of such techniques must be weighed against the potential risk from such modifications. While specific problems have not been reported, it is not difficult to imagine how a safety pin could be aspirated, or a non-deformable endotracheal tube connector might cause a pressure injury to the nares if left in place for more than a brief period.
Practice Considerations
During airway management, the person performing the procedure is responsible for the equipment being used on the patient. In most circumstances, the limited number of devices in use makes this a relatively simple task. This case highlights two aspects that complicate the situation.
First, there was an unplanned change in airway management as a result of the procedure becoming more complex. Changes in airway management should trigger a reassessment of the patient and formulation of a new airway plan. It is difficult to ascertain when the NPA was lost into the nasopharynx of this patient, but the location of the NPA should have been assessed during the transition from MAC to a general anesthetic, and again at emergence from anesthesia and extubation (removal of the endotracheal tube).
Second, responsibility for the airway was shifted back and forth between the CRNA and the anesthesiologist during the case. It is well recognized that ineffective communication is the most frequently cited category of root causes of sentinel events in hospitals.(7) Communications during handoffs or transitions of care are of vital importance. In 2007, The Joint Commission made the implementation of a standardized approach to handoff communications a 2007 National Patient Safety Goal. At the time of this writing, no specific standard of care for handoffs has been established by the American Society of Anesthesiologists; in practice, anesthesiologists often discuss how the airway is being managed when they transition care. A structured handoff between the CRNA and the anesthesiologist may have prompted an inquiry as to the whereabouts of the airway device. In this situation, a full handoff may not have been done since the CRNA was not actually leaving the procedure. Nonetheless, some discussion regarding the NPA should have occurred prior to intubation, since the anesthesiologist was not in the operating room when it was originally placed. The handoff to the recovery nurse in the post-anesthesia care unit was also an opportunity to discover the missing NPA.
In this case, the providers probably were individually trying to provide the highest quality care possible. But the failure of "usual individual practice" to recognize the retained NPA illustrates the need for a more systems-based approach. As there is limited literature on systems-based solutions to retained or aspirated airway devices, we can potentially extrapolate from a similar but more widely studied situation: the management of sponges and instruments during surgical procedures.
Retained surgical foreign objects have been the focus of considerable study (8) (and a previous AHRQ WebM&M commentary). Although relatively uncommon, RFOs during surgery have significant implications for the patient, including morbidity from the RFO, loss of patient trust, and additional costs related to the medical and legal resolution of the case.
The traditional method of preventing RFOs in surgery has been to perform structured counts that occur multiple times during the procedure.(9) In two recent large studies, the sensitivities of these counts are only 35%-77%.(10,11) Radiography is used in some institutions (10) to look for RFOs; however, the cost-effectiveness of this strategy over time remains unproven. One analytic model estimates costs of $1.1-$1.4 million per RFO event prevented.(12)
Looking to the future, the search for a better solution may lead to the application of technologies more commonly used in commercial inventory control than the operating room. Use of bar-coded sponges (13), electronic article surveillance tags, and radiofrequency identification (RFID) tags (14) has been advocated to help keep track of devices used during invasive procedures. While promising, more time and experience are needed to determine whether these will be cost-effective solutions for preventing RFOs.
Given that airway device retention or aspiration is probably rarer than RFOs in surgery, it is difficult to argue that patients without symptoms or airway obstruction should have routine radiography after surgical procedures. But is there anything else we can learn from the experience and literature around RFOs to help improve the safety of airway devices? Bar coding and other inventory-control approaches are unproven and still require human action to work properly. Structured counts of airway devices would likely be impractical, given the often emergent nature of airway instrumentation.
Airway management is a vitally important aspect of perioperative care, and complications can have serious consequences. Handoffs during patient care create opportunities for missed communications. When handoffs coincide with airway management, the stakes are even higher. To prevent retained airway devices, handoff communications need to include a thorough accounting of invasive devices. The development of standardized tools or checklists to facilitate handoffs may help remind practitioners and reinforce a culture of consistent communication.
Take-Home Points
- Intubation and airway management are invasive procedures with significant associated risks.
- Good handoff communication helps to prevent errors. Structured handoffs should be considered to minimize forgotten items and to reinforce a culture of consistency in handoff communications.
- Prevention of RFOs requires personal vigilance as well as systems approaches, though the optimal approach has not been determined—and probably will vary depending on the specific setting. The translation of inventory-control devices into the operative setting may prove useful, although this implementation is not mature.
Christopher R. Lee, MD Acting Assistant Professor, Department of Anesthesiology and Pain Medicine University of Washington School of Medicine Chair, Coordinated Quality Improvement Program
University of Washington Medical Center Department of Anesthesiology
References
1. Dhar V, Al-Reefy H, Dilkes M. Case report—an iatrogenic foreign body in the airway. Int J Surg. 2008;6:e46-e47. [go to PubMed]
2. Hayes JD, Lockrem JD. Aspiration of a nasal airway: a case report and principles of management. Anesthesiology. 1985;62:534-535. [go to PubMed]
3. Dua K, Saxena KN, Dua CK. Airway within airway: a case report. Indian J Anaesth. 2004;48:486-487. [Available at]
4. Milam MG, Miller KS. Aspiration of an artificial nasopharyngeal airway. Chest. 1988;93:223-224. [go to PubMed]
5. Mobbs AP. Retained nasopharyngeal airway. Reply. Anaesthesia. 1989;44:447. [Available at]
6. Mahajan R, Kumar S, Gupta R. Prevention of aspiration of nasopharyngeal airway. Anesth Analg. 2007;104:1313. [go to PubMed]
7. The Joint Commission. 2007 Hospital/Critical Access Hospital National Patient Safety Goals. [Available at]
8. Gawande AA, Studdert DM, Orav EJ, Brennan TA, Zinner MJ. Risk factors for retained instruments and sponges after surgery. N Engl J Med. 2003;348:229-235. [go to PubMed]
9. Recommended practices for sponge, sharps, and instrument counts. AORN J. 2006;83:418-433. [Available at]
10. Cima RR, Kollengode A, Garnatz J, Storsveen A, Weisbrod C, Deschamps C. Incidence and characteristics of potential and actual retained foreign object events in surgical patients. J Am Coll Surg. 2008;207:80-87. [go to PubMed]
11. Egorova NN, Moskowitz A, Gelijns A, et al. Managing the prevention of retained surgical instruments: what is the value of counting? Ann Surg. 2008;247:13-18. [go to PubMed]
12. Regenbogen SE, Greenberg CC, Resch SC, et al. Prevention of retained surgical sponges: a decision-analytic model predicting relative cost-effectiveness. Surgery. 2009;145:527-535. [go to PubMed]
13. Greenberg CC, Diaz-Flores R, Lipsitz SR, et al. Bar-coding surgical sponges to improve safety: a randomized controlled trial. Ann Surg. 2008;247:612-616. [go to PubMed]
14. Radio-frequency surgical sponge detection: a new way to lower the odds of leaving sponges (and similar items) in patients. Health Devices. 2008;37:193-202. [go to PubMed]
Figures
Figure 1a. Proper placement of nasopharyngeal airway device, a soft plastic tube inserted into the nose that extends into the posterior pharynx. (Illustrations by Chris Gralapp.)
Figure 1b. Illustration representing position of dislodged nasopharyngeal airway device. (Illustrations by Chris Gralapp.)
Illustrations by Chris Gralapp.