Bad Writing, Wrong Medication
Case Objectives
- Differentiate between a medication error and an adverse drug event.
- Appreciate the system complexities involved in medication errors that occur in ambulatory settings.
- List at least three to five elements that comprise a framework for conducting a root cause analysis of a medication error.
Case & Commentary: Part 1
A 73-year-old man with a long-standing cardiac arrhythmia came to the ambulatory clinic for a routine follow-up visit. After evaluation, he received a handwritten prescription for Rythmol (propafenone), 150 mg, which had been his usual antiarrhythmic medication for the past 3 years. The patient delivered the prescription to the clinic pharmacy, and it was filled. Shortly after starting to take the medication, the man began to feel "very, very bad," with nausea, sweating, and an irregular heartbeat. These symptoms persisted for 2 weeks, and the patient called his physician to schedule another appointment. The patient brought the medication to his physician, stating that the Rythmol tablets looked different from their usual appearance.
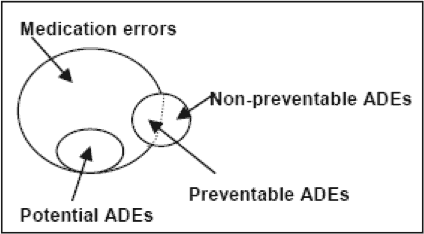
Both medication errors and adverse drug events (ADEs) are types of medication safety events. To provide a framework for discussion, it is useful to begin with a definition of each. The National Coordinating Council on Medication Error Reporting and Prevention defines a medication error as "any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the health care professional, patient, or consumer."(1) Such events may be related to any aspect of medication use. An ADE is defined as "an injury resulting from a medical intervention related to a drug."(2) The relationship between the two is illustrated in the Figure. Medication errors that cause harm become preventable ADEs; errors that do not cause harm are termed potential ADEs. Some ADEs are not due to errors and are not preventable.
Research that characterizes the epidemiology of medication errors and ADEs in the ambulatory setting is sparse, both due to the fragmented nature of most outpatient care and the limitations of the relevant data sources. Nonetheless, it is estimated that 21% of handwritten prescriptions contain at least one prescription-writing error.(3) Most research has focused on dispensing errors, and investigators estimate that between 1.7% and 24% of prescriptions are dispensed erroneously, and that 1.5% to 4% of these errors could cause harm.(4) Labeling errors and wrong-drug errors occur frequently.(4,5) Investigators estimate that between 11% and 38% of ADEs in the ambulatory setting are preventable (6-9), and one study found that preventable ADEs were more likely than nonpreventable ADEs to be serious, life-threatening, or fatal (54% versus 30%, respectively).(9) They further estimated the direct cost of a preventable ADE to be $1983 in older adults, and the estimated annual direct cost of preventable ADEs to the entire Medicare population to be $887 million (both 2000 $). These costs were attributed to inpatient stays (62%), emergency department visits (6%), outpatient care (28%), and medications (4%). These estimates do not include costs to the patient, productivity losses, or decrements in quality of life.(9)
Data from the 2006 National Ambulatory Medical Care Survey indicate that one or more drugs were provided or prescribed in 636.7 million of the 902 million office visits made to physicians in 2006.(10) With such high prescription volumes, adopting strategies to optimize medication safety in the ambulatory setting is a crucial component of improving the quality and safety of patient care.
In this case, the patient's statement that the physical characteristics of the dispensed medication differed from those of his usual prescription, coupled with the temporal association between prescription filling and his symptoms of nausea, sweating and irregular heartbeat, send a clear signal that a dispensing error likely occurred.
Case & Commentary: Part 2
Based upon the altered appearance of the tablets, both the patient and the physician suspected that this might not be the correct drug. Upon investigation, the physician identified the patient's medication as Synthroid (levothyroxine), 150 mcg, not the intended Rythmol (propafenone), 150 mg. When the physician spoke with the pharmacist who had filled the prescription, it became apparent that a medication dispensing error had occurred due to unclear handwriting on the original prescription. The patient's symptoms of nausea, sweating, and irregular heartbeat were related to both inadvertent, abrupt discontinuation of Rythmol and the unnecessary use of Synthroid at a relatively high initial dosage. Synthroid was immediately discontinued, and the patient restarted Rythmol as originally prescribed.
In a systems-based approach to medical error prevention, we recognize that many errors stem from weaknesses in individual components of the health care system, that these weaknesses are best improved through multidisciplinary efforts, and that determining who was involved is less important than identifying and addressing the root causes of the problem.(11) The Institute for Safe Medication Practices (ISMP) actively promotes this approach and has conducted hundreds of root cause analyses (RCAs) after error occurrences. In all cases, the causes have been multifactorial and have involved many processes, more than one line of responsibility, and organization-wide systems.(11) ISMP has identified ten key system elements that influence medication safety: (i) access to patient-specific information; (ii) access to drug information; (iii) adequate communication; (iv) proper labeling, packaging, and nomenclature; (v) drug standardization, storage, and monitoring; (vi) medication delivery device use and monitoring; (vii) sufficient environmental support; (viii) staff competency and education; (ix) patient education; and (x) quality improvement and risk management programs.
Much is made of the roles of electronic medication records (EMRs), computerized provider order entry (CPOE) systems, and bar-coding technologies in reducing errors. The Institute of Medicine strongly supports greater use of information technology to improve medication safety.(12) CPOE systems, when correctly implemented, are associated with a reduction in errors—in some studies by more than 50%.(13,14) In a recent study that investigated the impact of a CPOE system on errors in the ambulatory clinic setting, the greatest reduction was seen in the odds of an error of illegibility (97% reduction), followed by use of inappropriate abbreviations (94%), and missing information (85%).(14) The literature is silent on the impact of bar-coding in reducing medication errors in the community pharmacy setting.
Recent survey data from the National Center for Health Statistics indicate that approximately 17% of office-based physicians used a "basic" EMR in 2008 and 4% used a "fully functional" system.(15) As defined, a basic system included functionality for prescription ordering, while a fully functional system also allowed for electronic prescription transmittal. When prescriptions are electronically prescribed, they cannot always be transmitted directly to the pharmacy dispensing computer system, but may require that the prescription be sent via facsimile. Pharmacies can rescan these into the pharmacy system for documentation purposes, but they must still re-enter the prescription data into the pharmacy computer system to dispense the medication(s). Even when direct transmittal is successful, routed and delivered through an interchange used by the pharmacy benefit manager, the prescription often requires modification to ready it for dispensing. Finally, community pharmacy computer systems are seldom linked to EMRs and CPOE systems. At a minimum, pharmacy dispensing computer systems do contain a record of patient demographic information, allergies, and a list of prescriptions filled at that pharmacy. They provide alert checking for allergies, duplicate medications, and drug–drug interactions. Virtually all pharmacies have access to drug information with online information being more up-to-date than paper versions.
Pharmacists are accustomed to correctly interpreting handwritten prescriptions. When illegibility raises doubt in a pharmacist's mind, the pharmacist may seek clarification by viewing the EMR, checking with the patient, or telephoning the prescriber's office. Once clarified, bar-coding can be used to match each unique prescription to the drug product. Once a prescription is filled, the purchase transaction is often handled by a pharmacy assistant or technician. The Omnibus Budget Reconciliation Act of 1990 required states to establish standards governing patient counseling for prescriptions.(16) Most states require the offer of counseling, which is usually made by this same technician. If a patient accepts, face-to-face counseling by a pharmacist is required; in 64% of states for new prescriptions only, in the remainder for both new prescriptions and refills. This interchange provides the opportunity for a final check to prevent medication errors. Yet patients often refuse counseling, especially for medication refills. In one recent study, 43 of 100 trained shoppers received verbal counseling for a new prescription. In 16 of the cases, the shopper prompted counseling. All shoppers received written information covering an average of 90% of required topics.(17) In a similar study, 63% of trained shoppers received verbal counseling.(18) Characteristics associated with more frequent counseling were younger pharmacists, increased intensity of state counseling requirements, and less busy pharmacies.
Historically, the medication error described in this case might have been dismissed solely as a dispensing error caused by an illegible prescription. Conducting an RCA, using ISMP's ten key system elements as a framework, can shed light on the underlying causes of the error, and on improvements that could be made to mitigate chances of recurrence.(19) First, the pharmacist may have lacked adequate patient-specific information with which to confirm the correct medication. It appears that the intended purpose was not written on the prescription. The ISMP advocates that the intended use be indicated on each prescription as an aid for pharmacists and for patients.(11) A CPOE system was not used to prescribe or to transmit the prescription. The pharmacy was associated with the clinic, but we do not know whether the clinic had an EMR in place, and whether the pharmacist could access it. Second, difficulties with interprofessional communication may have discouraged the pharmacist from calling to seek clarification. Third, the pharmacist may never have doubted that the prescription was for Synthroid. Drug names that look alike are easily confused, and a mix-up between Rythmol and Synthroid is plausible. Fourth, perhaps the physician and/or pharmacist felt rushed in their environments due to competing priorities, staffing challenges, or an impatient patient. Poor lighting, inefficient workflow, or interruptions may have contributed. Finally, we do not know if the patient was offered counseling for the "new" medication. If it was offered, he may have refused it, thinking that his prescription was for his usual refill. Rythmol 150-mg tablets are white, round, and scored; Synthroid 150-mcg tablets are light blue, round, partially scored, and smaller.(20) The two medications looked dissimilar, and the error could have been corrected sooner had the patient questioned the pharmacist at the time of dispensing or shortly thereafter.
In this case, the medication error, a preventable ADE, led to patient harm. Implementing the necessary system improvements could minimize recurrence of this type of error. Most importantly, implementation of a CPOE system would have eliminated prescription illegibility, the precipitating factor in this case. In addition, three indirect solutions could contribute to error avoidance: (i) pharmacist access to patient-specific information in an EMR, or at least the intended purpose listed on the prescription; (ii) patient counseling at the point of dispensing; and (iii) patient empowerment to question the change in prescription in a timely fashion. Finally, if communication or environmental issues were implicated, these should be addressed.
Health care systems are adopting systems approaches to improve patient safety. This, coupled with the incentives spurring adoption of EMRs and CPOE systems and promoting their interoperability among settings (21), should result in a marked reduction of this type of error over time.
Take-Home Points
- Medication errors are potential adverse drug events; medication errors that cause harm are preventable adverse drug events. Not all adverse drug events are caused by medication errors.
- Because outpatient ambulatory health care delivery is a fragmented process, estimating medication errors and adverse drug events in this setting is challenging.
- Even seemingly simple medication errors are multifactorial, involving more than one process and more than one line of responsibility.
- As with medication errors that occur in acute care environments, serious ambulatory medication errors should also be analyzed from a systems perspective (root cause analysis) by members of an interdisciplinary team.
- Electronic medical records with computerized provider order entry systems, when implemented correctly, will mitigate certain types of medication errors, thereby improving medication safety.
Beth Devine, PharmD, MBA, PhD Research Associate Professor Pharmaceutical Outcomes Research & Policy Program, School of Pharmacy
Biomedical & Health Informatics, School of Medicine
University of Washington
Faculty Disclosure: Dr. Devine declares that she has no financial arrangements or other relationship with the manufacturers of any commercial products discussed in this continuing education activity. In addition, her commentary does not include information regarding investigational or off-label use of pharmaceutical products or medical devices.
References
1. National Coordinating Council for Medication Error and Reporting and Prevention. [Available at]
2. Bates DW, Boyle DL, Vander Vliet MB, Schneider J, Leape L. Relationship between medication errors and adverse drug events. J Gen Intern Med. 1995;10:199-205. [go to PubMed]
3. Shaughnessey AF, Nickel RO. Prescription-writing patterns and errors in a family medicine residency program. J Fam Pract. 1989;29:290-295. [go to PubMed]
4. Flynn EA, Barker KN. Research on errors in dispensing and medication administration. In: Cohen MR, ed. Medication Errors, 2nd ed. Washington, DC: American Pharmacists Association; 2007:20. ISBN: 9781582120928.
5. Rolland P. Occurrence of dispensing errors and efforts to reduce medication errors at the Central Arkansas Veteran's Healthcare System. Drug Saf. 2004;27:271-282. [go to PubMed]
6. Honigman B, Lee J, Rothschild J, et al. Using computerized data to identify adverse drug events in outpatients. J Am Med Inform Assoc. 2001;8:254-266. [go to PubMed]
7. Gandhi TK, Weingart SN, Borus J, et al. Adverse drug events in ambulatory care. N Engl J Med. 2003;348:1556-1564. [go to PubMed]
8. Gurwitz JH, Field TS, Harrold LR, et al. Incidence and preventability of adverse drug events among older persons in the ambulatory setting. JAMA. 2003;289:1107-1116. [go to PubMed]
9. Field TS, Gilman BH, Subramanian S, Fuller JC, Bates DW, Gurwitz JH. The costs associated with adverse drug events among older adults in the ambulatory setting. Med Care. 2005;43:1171-1176. [go to PubMed]
10. Cherry DK, Hing E, Woodwell DA, Rechtsteiner EA. National Ambulatory Medical Care Survey: 2006 summary. Natl Health Stat Report. 2008:1-39. [go to PubMed]
11. Cohen MR. Causes of medication errors. In: Cohen MR, ed. Medication Errors, 2nd ed. Washington, DC: American Pharmacists Association; 2007:55-66. ISBN: 9781582120928.
12. Aspden P, Wolcott J, Bootman JL, Cronenwett LR, eds. Preventing Medication Errors: Quality Chasm Series. Washington, DC: National Academies Press; 2006. ISBN: 9780309101479.
13. Bates DW, Leape LL, Cullen DJ, et al. Effect of computerized physician order entry and a team intervention on prevention of serious medication errors. JAMA. 1998;280:1311-1316. [go to PubMed]
14. Devine EB, Hansen RN, Wilson-Norton JL, et al. The impact of computerized provider order entry on medication errors in a multispecialty group practice. J Am Med Inform Assoc. 2010;17:78-84. [go to PubMed]
15. Hsiao CJ, Beatty PC, Hing ES, Woodwell DA, Rechtsteiner EA, Sisk JE. Electronic medical record/electronic health record use by office-based physicians: United States, 2008 and Preliminary 2009. National Center for Health Statistics. December 2009. [Available at]
16. Vivian JC, Fink JL. OBRA '90 at Sweet Sixteen: A retrospective review. US Pharmacist 2008;33:59-65. [Available at]
17. Flynn EA, Barker KN, Berger BA, et al. Dispensing errors and counseling quality in 100 pharmacies. J Am Pharm Assoc (2003). 2009;49:171-180. [go to PubMed]
18. Svarstad BL, Bultman DC, Mount JK. Patient counseling provided in community pharmacies: effects of state regulation, pharmacist age, and busyness. J Am Pharm Assoc (2003). 2004;44:22-29. [go to PubMed]
19. Burkhardt M, Lee C, Taylor L, Williams R, Bagian J. Root cause analysis of medication errors. In: Cohen MR, ed. Medication Errors. 2nd ed. Washington, DC: American Pharmacists Association; 2007:67. ISBN: 9781582120928.
20. Drug Identifier [database online]. Facts & Comparisons e-Answers. New York, NY: Wolters Kluwer Health; 2010. [Available at]
21. Blumenthal D. Stimulating the adoption of health information technology. N Engl J Med 2009;360:1477-1479. [go to PubMed]
Figure
Figure. Relationship between medication errors and adverse drug events.(2)