The Dose Makes the Poison: Medication Error During Procedural Sedation in the Pediatric Emergency Department.
Amashta ML, Barnes DK. The Dose Makes the Poison: Medication Error During Procedural Sedation in the Pediatric Emergency Department.. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2023.
Amashta ML, Barnes DK. The Dose Makes the Poison: Medication Error During Procedural Sedation in the Pediatric Emergency Department.. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2023.
Patrick Romano, MD, MPH; Debra Bakerjian, PhD, APRN, RN; Michael Leonardo Amashta, MD; David Barnes, MD; Patricia Poole, PharmD for this Spotlight Case and Commentary have disclosed no relevant financial relationships with ineligible companies related to this CME activity
Learning Objectives
- Discuss the purpose and benefits of procedural sedation performed in non-surgical settings.
- Describe the four levels of procedural sedation.
- Understand common indications and risks associated with procedural sedation.
- Review commonly used medications for procedural sedation to include sedative hypnotics, analgesics, inhalational, and dissociative amnestic agents.
- Implement procedural sedation best practices including use of adequate personnel, continuous monitoring, and use of checklists.
- Appreciate the importance of standardized medication labeling and dosing, closed loop communication, and medication administration rights in preventing medication errors.
- Recognize barriers to routine medication near miss and error reporting.
The Case
A three-year-old girl weighing 20 kilograms (kg) was transferred by ambulance to a tertiary Emergency Department (ED) from a referring hospital due to a left leg injury. She reportedly slipped while running on a wet floor. Imaging performed at the transferring facility indicated “left posterior hip dislocation vs. pathological fracture” and it was determined she needed pediatric orthopedics specialty care. Initial vital signs were notable for tachycardia and tachypnea, but these findings normalized after the parents calmed her. The patient was crying and holding her left hip flexed with significant tenderness on examination, but there were no other signs of trauma. She was given intravenous morphine for pain control and started on maintenance intravenous fluids. Imaging demonstrated that the left femoral head was smaller than the right with premature ossification of the femoral head physis (likely reflecting developmental dysplasia of the hip), now complicated by left femoral-acetabular dislocation. There was no acute fracture and no acute findings involving the tibia or fibula. An orthopedic surgeon was consulted and recommended closed reduction of the left hip in the ED.
The ED physician obtained informed consent for procedural sedation from the parents after full discussion of procedural risks and benefits. After an appropriate procedural pause, nursing staff applied cardiac, pulse oximetry, and end tidal carbon dioxide monitors. With the orthopedic and ED teams at the bedside, the patient was pre-oxygenated with high-flow oxygen. Ketamine 20 mg (1 mg/kg) was administered intravenously. Following the ED’s standard protocol for procedural sedation, the ED resident physician then called out to the scribing nurse the dose of propofol they would administer; 10 mg was the intended dose. The resident administered the medication and called out “10 of propofol”. The nurse repeated “10 of propofol given.” The orthopedic surgeon performed the hip reduction successfully.
The patient became apneic shortly thereafter. She was easily ventilated for one minute with jaw thrust and bag-valve-mask. She regained consciousness and was able to maintain her airway and breathe spontaneously. Her end tidal CO2 normalized, and she remained hemodynamically stable. The patient continued to recover and was monitored appropriately in the intensive care unit. Subsequent case review confirmed that the resident administered 10 mL of a 10 mg/mL solution of propofol, a total dose of 100 mg (5 mg/kg), instead of the intended volume of 1 mL of a 10 mg/mL solution, a total dose of 10 mg (0.5 mg/kg).
The Commentary
By Michael Leonardo Amashta, MD, and David K. Barnes, MD,
Background
As clinician experience and expertise evolve, the frequency of pediatric procedural sedation performed outside the operating room continues to increase.1 Pediatric procedural sedation is now commonly performed in the Emergency Department (ED), inpatient units, and ambulatory locations such as outpatient physician offices and imaging centers.
Procedural sedation in the pediatric population is a high-risk intervention. Pediatric patients are more likely to experience respiratory depression and life-threatening hypoxia than healthy adults because of their unique airway anatomy and respiratory physiology.2,3 A comprehensive study by the Pediatric Sedation Research Consortium comprising more than 30,000 cases of pediatric procedural sedation performed outside the operating room reported a 5.3% incidence of complications. However, the incidence of adverse events requiring hospital admission was only 1 in 1500 cases. Only one case required cardiopulmonary resuscitation and no deaths were reported.3
This case is an example of a medication error related to pediatric procedural sedation. Specifically, it a highlights the value of implementing system-wide safety protocols and practices to prevent medication administration errors during high-risk procedures. Previous work describing nurse-related medication administration errors has emphasized the complexity of weight-based dosing as a significant contributing factor to medication errors in pediatric populations. Medication administration is a complex process with each step being vulnerable to error. Risk factors and prevention techniques to reduce the risk of adverse drug events have been previously described. This commentary will review pediatric procedural sedation best practices, including those associated with medication safety.
Principles of Procedural Sedation
The purpose and benefits of procedural sedation
The administration of analgesic, anxiolytic, and hypnotic (sedative) medications is frequently required to facilitate painful and/or anxiety provoking medical tests or procedures in various clinical settings. The American College of Emergency Physicians (ACEP) adopted a formal definition of procedural sedation: “the use of anxiolytic, sedative, hypnotic, analgesic, and/or dissociative medication(s) to attenuate anxiety, pain, and/or motion. These agents are administered to facilitate amnesia or decreased awareness and/or patient comfort and safety during a diagnostic or therapeutic procedure.”4,5 Sedation can also ameliorate the psychological trauma associated with procedural stress, especially in pediatric patients who are often frightened, lack coping skills, and are prone to experience negative emotional reactions to strangers and pain.6
Levels of sedation
Nearly all sedation scales incorporate assessment of patient alertness, respiratory effort, airway patency, and hemodynamic stability. Some also assess patient movement.4,5,7,8 Although routinely categorized into one of four levels, sedation depth should be conceptualized as a continuum ranging from normal level of alertness to complete unconsciousness, rather than as distinct categories with clearly defined boundaries and transitions. The level of sedation for any procedure should be selected based upon the procedure being performed, its expected duration, and the patient’s comorbidities and risk factors. The four most common levels of sedation used are:4
- Minimal Sedation/Anxiolysis: “A drug-induced state during which patients respond normally to verbal commands. Although cognitive function and coordination might be impaired, ventilatory and cardiovascular functions are unaffected.”
- Moderate Sedation:A drug-induced depression of consciousness during which patients respond purposefully to verbal commands, either alone or accompanied by light tactile stimulation. No interventions are required to maintain a patent airway, and spontaneous ventilation is adequate. Cardiovascular function is usually maintained.” Dissociative Sedation is a form of moderate sedation described as: “A trance-like cataleptic state induced by the dissociative drug ketamine characterized by profound analgesia and amnesia, with retention of protective airway reflexes, spontaneous respirations, and cardiopulmonary stability
- Deep Sedation: “A drug-induced depression of consciousness during which patients cannot be easily aroused but respond purposefully following repeated or painful stimulation. The ability to independently maintain ventilatory function may be impaired. Patients may require assistance in maintaining a patent airway and spontaneous ventilation may be inadequate. Cardiovascular function is usually maintained.
- General Anesthesia: “A drug-induced loss of consciousness during which patients are not arousable, even by painful stimulation. The ability to independently maintain ventilatory function is often impaired. Patients often require assistance in maintaining a patent airway, and positive pressure ventilation may be required because of depressed spontaneous ventilation or drug-induced depression of neuromuscular function. Cardiovascular function may be impaired.”4
Risks associated with procedural sedation
Although procedural sedation is considered a safe and effective practice when performed by experienced providers and healthcare teams, well-known risks exist. Complications associated with procedural sedation include apnea, hypoxia, vomiting, aspiration, laryngospasm, cardiovascular collapse, and adverse behavioral reactions (e.g., emergence reaction). Factors such as the patient’s anatomy, physiology, age, past medical history, allergies, physical status, intended depth of sedation, and side effect profiles of sedation agents, contribute to the overall risk of such adverse events. The American Society of Anesthesiologists (ASA) Physical Status Classification System was created in 1962 to assess patients’ pre-anesthesia medical co-morbidities and thereby to define levels of perioperative risk of morbidity and mortality.7 A prospective observational database of community EDs noted an increased rate of complications in patients with higher ASA classifications and with deeper levels of procedural sedation.9
Indications for procedural sedation
Procedural sedation is commonly used in patients of all ages, without regard to timing of last oral intake, to facilitate painful procedures such as fracture and dislocation reduction, hernia reduction, burn debridement, and cardioversion. In addition to these procedures, pediatric procedural sedation is commonly used for eye examination, laceration repair, abscess incision and drainage, intravenous access, and acquisition of imaging studies such as magnetic resonance imaging (MRI).5,10
Commonly used procedural sedation agents
The selection of sedation agents depends heavily on provider experience and preference. Drug selection should be tailored to each specific patient encounter with consideration given to drug allergies, current cardiopulmonary status, co-administered medications, and the patient’s prior experiences with procedural sedation, if applicable. Proceduralist preferences may also play a role in the selection of sedation agents. General classes of medications utilized in procedural sedation include sedative hypnotics, analgesics, inhalational agents, and dissociative amnestic agents.
Benzodiazepines such as midazolam are useful and widely preferred agents when anxiolysis is the primary goal of procedural sedation. Despite its sedative and anxiolytic effects, midazolam may paradoxically precipitate agitation in children.11,12 Propofol is an effective sedative due to its many appealing characteristics: rapid onset of action and combined sedative, amnestic, and antiemetic properties. Propofol does not have analgesic properties and is therefore typically co-administered with opioids or ketamine for painful procedures. The use of ketamine for procedural sedation is common, especially in pediatric populations, due to its favorable safety profile and dissociative and analgesic properties.10,13 Etomidate is a fast-acting sedative hypnotic with no analgesic properties and low reported rates of hypoxia and hypotension.10 Nitrous oxide, a commonly used inhalational agent with rapid onset of action, provides mild anxiolysis, sedation, and analgesia. At concentrations that range 30%-70%, it is typically blended with oxygen and can be self-administered by patients.8,10,14 When administered as the sole agent and at concentrations below 50%, nitrous oxide produces only minimal sedation. It is therefore administered with analgesics such as opioids.5,14
The coadministration of ketamine and propofol in lower than typical doses—a mixture commonly known as “ketofol”—is preferred by some providers due to the drugs’ synergistic effects and favorable side effect profile compared to standard doses of each. A meta-analysis comparing the analgesic properties and side effects of ketofol compared to propofol alone in adult procedural sedation found decreased rates of respiratory, cardiovascular, and bradycardic complications with ketofol.15 In contrast, the combination of ketamine and propofol has been associated with increased adverse events in pediatric procedural sedation compared with ketamine alone.10,16,17
Approach to Improving Safety & Patient Safety Target
Procedural sedation is ubiquitous in modern clinical medicine. Given the risks inherent to the practice, it is incumbent upon all those who perform sedation or monitor sedated patients to implement strategies and techniques demonstrated to improve safety and reduce the risk of adverse events and unfavorable patient outcomes
Two-person technique
The adage that there is safety in numbers is true for procedural sedation. However, the extent to which patients are attended and monitored during procedural sedation varies substantially across clinical areas and institutions. Workplace culture and human resource availability are common barriers to best practices
To understand these differences, it is useful to define the usual and customary roles assigned during procedural sedation. The sedation provider is responsible for administering sedation agents and directly monitoring the patient. This person should have experience and expertise with procedural sedation and emergency airway management. They may be an independent provider such as a physician or anesthetist, or a nurse or respiratory therapist executing physician orders. The proceduralist is the provider responsible for the primary procedure being performed, but they may also direct others to administer sedation on their behalf. They may therefore serve as both sedation provider and proceduralist simultaneously. Their attention and focus on performing the primary procedure (e.g., fracture reduction) may create a patient safety hazard. A third provider, most commonly a nurse, supports the sedation provider and proceduralist by documenting, monitoring, and executing tasks.4,8
It is unsafe to administer procedural sedation without adequate monitoring and supervision. In fact, both the American Academy of Pediatrics (AAP) and American College of Emergency Physicians (ACEP) recommend a minimum of two healthcare personnel at the bedside during procedures requiring sedation. Some guidelines recommend three personnel at bedside—the proceduralist, the sedation provider, and a support staff person—when deep sedation is targeted.8 However, various studies have demonstrated safety and effectiveness with a single physician administering moderate or deep sedation and performing the procedure while a nurse monitors the patient.18,19 We recommend employing the three-provider model for all procedural sedation cases if resources allow and personnel are available, especially when deep sedation is targeted. However, circumstances may exist when sedation is urgently needed to prevent harm associated with the underlying condition or injury but a third provider is unavailable.18
Pre-procedure checklist
A pre-procedure checklist is a simple and effective tool that can reduce the risk of adverse events related to procedural sedation. Checklists, used in many non-healthcare settings such as commercial aviation, have been shown to improve safety and have been increasingly used in healthcare.20 One well-studied example of a patient care checklist is the World Health Organization (WHO) surgical safety checklist, which has been implemented globally and is associated with decreased rates of surgical site infections and surgical complications.21 While it may seem intuitive that checklists designed to prevent patient safety lapses and reduce cognitive errors would demonstrate conclusive benefit, studies evaluating their effectiveness in pediatric populations undergoing procedural sedation show mixed results.
In one study, a pediatric-specific, modified WHO Procedural Sedation Safety Checklist incorporating critical safety elements (CSEs), such as equipment check, medication double check, and use of a timeout, reduced both moderate (e.g., laryngospasm, airway obstruction, apnea, nonminimal hypoxia) and severe (e.g., aspiration, cardiac arrest, unplanned intubation, death) adverse events at a free-standing children’s hospital. Specifically, the study showed completion of four CSEs included in the safety checklist was associated with reduced rates of adverse events in patients in the ED, pediatric intensive care unit, and children’s ward. Additionally, an adverse event was reported with every instance in which a medication was dosed greater than the recommended maximum. Although the rate of adverse events decreased by 30% after the checklist was implemented, results lacked statistical significance likely due to low baseline adverse event rates.22
Implementation of a similar pre-sedation checklist in a pediatric ED population sedated with ketamine and propofol failed to reduce serious adverse events (SAEs). A total of 183/1349 (13.5%) SAEs occurred in the pre-checklist group while 420/1846 (22.7%) SAEs occurred in the post-checklist group (p<0.0001), an increased rate of SAEs. Specifically, rates of laryngospasm (0.6% versus 1.6%, p<0.05), apnea (3.5% versus 4.2%, p=0.37), and hypoxia (SpO2 <90%) (9.1% versus 16.9%, p<0.0001) were all higher in the post-checklist group. Fortunately, all procedures were successfully completed, and no patients required hospitalization because of a SAE. 23
Equipment and resource preparation
Procedural sedation requires not just specialized monitoring equipment but also preparation and availability of airway management and resuscitation equipment should the patient have an unexpected adverse event during the procedure. The easy-to-recall “SOAP ME” mnemonic is commonly used before invasive airway procedures, such as endotracheal intubation, and can also be used before procedural sedation to gather and prepare necessary equipment and resources.8
S = Size-appropriate suction catheters and a functioning suction apparatus (e.g., Yankauer-type suction)
O = an adequate Oxygen supply and functioning flow meters or other devices to allow its delivery
A = size-appropriate Airway equipment: bag-valve-mask or equivalent device [functioning]), nasopharyngeal and oropharyngeal airways, laryngeal mask airway, laryngoscope blades (checked and functioning), endotracheal tubes, stylets, face mask
P = Pharmacy: all the basic drugs needed to support life during an emergency, including antagonists as indicated
M = Monitors: functioning pulse oximeter with size-appropriate oximeter probes, end-tidal carbon dioxide monitor, and other monitors as appropriate for the procedure (e.g., noninvasive blood pressure, ECG, stethoscope)
E = special Equipment or drugs for a particular case (e.g., defibrillator)
Monitoring during procedural sedation
Routine cardiac monitoring equipment should always be utilized during procedural sedation. Blood pressure should be measured frequently, and the electrocardiogram, heart rate, respiratory rate, pulse oximetry, and capnography (i.e., end-tidal carbon dioxide) should be continuously monitored and documented every 5-10 minutes.4,5,8,10
Both ACEP and AAP recommend the use of continuous waveform capnography during moderate and deep sedation.4,8 The use of waveform capnography—which differs from capnometry in which only the quantitative end-tidal carbon dioxide (CO2) value is displayed—allows clinicians to quickly and effectively detect hypoventilation, airway obstruction, and apnea. Capnography can detect hypoventilation caused by decreased respiratory drive leading to a rise in end-tidal CO2 levels above 50 mmHg. Capnography can also detect hypoventilation due to decreased tidal volume with a relatively normal respiratory rate, suggested by end-tidal CO2 levels less than 30 mmHg or a decrease greater than 10 mmHg from the steady state level.10,24 A prospective observational study in ED patents demonstrated the value of continuous nasal waveform capnography. Continuous pulse oximetry detected only a third of patients who met criteria for respiratory depression whereas capnography detected all patients with respiratory depression.24
ACEP recommends the use of supplemental oxygen prior to and during procedural sedation. The intervention delays time to desaturation and allows patients to safely tolerate short periods of hypoventilation by increasing oxygen reserves without the potential harms of positive pressure ventilation.4 Supplemental oxygen is not recommended without capnography because the detection of hypoxemia can be delayed.4,5,10
Nine rights of medication administration
This case highlights the importance of appropriate provider experience and training to prevent adverse events related to medication administration resulting in patient harm. Nurses learn the “Five Rights of Medication Administration”25 but some experts propose that all providers who administer medications should learn and abide by the “Nine Medication Administration Rights”.26 These “nine rights” are:
- Right patient
- Right drug
- Right route
- Right time
- Right dose
- Right documentation
- Right action
- Right form (or formulation)
- Right response
The right action requires providers who administer or prescribe medications to ensure the correct clinical indication. For example, potassium supplements for a patient on furosemide is routine, however, this same drug may lead to life-threatening consequences in a patient who is hyperkalemic. This case involved at least one medication administration right that was not followed: right dose.
Standardized medication dosing and labeling
As noted in the Joint Commission’s Sentinel Event Alert “Preventing Pediatric Medication Errors”, adverse drug events occur three times more frequently in pediatric patients. Several factors explain why children are more susceptible to medication errors that result in harm, the most important being the requirement for weight-based dosing. Most medications are formulated and packaged for adults. Sedation providers may fail to recognize that pediatric weight-based dosing involves handling medications formulated with variable concentrations requiring careful attention to administer the correct dose. Moreover, children often lack the ability to communicate that they are experiencing an adverse or unintended effect from a medication.27 A systematic review of medication errors in pediatric patients found the most common type of medication error was an incorrect dosing error frequently involving a dosage ten times that intended, similar to the present case.28
To prevent dose-related errors, high-risk medications should be available in limited concentrations and doses. Standard dosing units (e.g., mg/mL) should always be used, and appropriate size syringes should be tailored to the medication dose and volume. For example, a toddler weighing 10 kg who should receive 1 mg/kg of ketamine at a concentration of 10 mg/mL should receive a volume of 1 mL in the smallest syringe possible to minimize the risk of a dosing error if a larger syringe were used.27,29,30
Individualized doses in pre-filled, single-use syringes can reduce the risk of a medication overdose. The Institute for Safe Medication Practices (ISMP) defines these “ready-to-administer" (RTA) prefilled syringes as “an injectable product containing the active drug in solution at the required concentration and volume, presented in the final container (syringe, infusion bag, or elastomeric device) and ready to be administered to the patient.”31 In two randomized controlled simulation studies, medical staff (nurses and physicians) were provided either prefilled medication syringes or allowed to follow standard medication protocols during simulated resuscitations. The group that used prefilled syringes made fewer medication errors and administered medications more quickly.32,33
The availability of a clinical pharmacist provides another important and useful resource when high-risk medications are administered. Clinical pharmacists can provide guidance and oversight throughout the medication ordering and administration processes.27,34 Preprinted dosage calculation sheets and computerized physician order entry templates for emergency medications, expressed in both mass and volume units, have been recommended for pediatric critical care settings and may have relevance in other settings.27
Syringe labeling, especially when the person preparing the medication is not the one administering it, can reduce medication errors. Medications that are not readily available in prefilled syringes and require preparation from a source vial should always be clearly and concisely labeled (preferably printed, not hand-written). This label should be applied to the syringe and should contain the name of the drug, drug concentration, and total dose. In the case of similar sounding and similarly spelled medications (e.g., Versed and vecuronium, or dexmedetomidine and dexamethasone), every effort should be made to prevent erroneous administration: distinct labeling, storage of easily-confused medications in separate locations, and medical staff training using resources such as the Joint Commission/ISMP list of confused drug names.34–38
The use of shared language and terminology—in this case dose units—is critical and related to the concept of standardized labeling. The resident physician’s assumed use of milliliter units did not align with the nurse’s assumption that milligram units would be used. Moreover, neither party called out the units they were using for the other to hear and acknowledge. In our opinion, this was a root cause of the error and suggests that both latent errors (i.e., related to system and product design such as how medications are manufactured and packaged) and active errors (i.e., slips, lapses, or mistakes made by the operators) occurred. The use of shared terminology is a critical component of closed loop communication.
Closed-loop communication—readback and hearback
Communication failure is commonly implicated as a key contributing factor to sentinel events and patient harm.39 One study that analyzed adverse events associated with adult procedural sedation outside the operating room reported that miscommunication rates were highest in the ED and in those areas with high patient volumes, greater number of consultants, and high stress.40
The Joint Commission supports the use of standardized methods of communication. Closed loop communication can be operationalized through the readback and hearback technique, which can be used in a variety of clinical scenarios including verbal orders for medication administration.39 Readbacks ensure verbal messages and orders are accurately received and understood by the receiver, whereas hearbacks close the loop with the sender. An ideal, closed-loop communication process is described in the following order:
- The sender concisely states information to the receiver.
- The receiver is then required to read back or say what was heard.
- The sender then provides a hearback, acknowledging that the readback was correct, or making a correction.
- The readback/hearback process continues until a shared understanding is mutually verified.39
There is a direct relationship between poor closed-loop communication and clinically relevant errors related to medications and equipment.41 Effective closed-loop communication requires deliberate practice, discipline, engagement, regular training, a system of accountability, and feedback. For example, an intervention to improve closed-loop communication in a cardiac catheterization suite via performance feedback and staff education demonstrated a significant increase in the use of closed-loop communication.42
Medication error reporting
The safest organizations learn from their mistakes. Unfortunately, medication errors are underreported, which limits the value of learning from those errors. Numerous barriers to reporting exist including fear of consequences, a culture of blame, perception of lack of benefit/action, inadequate time to report errors (given cumbersome reporting systems), and lack of knowledge about what to report.43,44 Better systems to report medication errors in a user-friendly and non-punitive manner are needed, as are clear reporting guidelines and cultures that embrace humility, personal growth, and organizational learning when errors are made. Yet even when a medication-related adverse event is reported, the lack of a common taxonomy and nomenclature makes systematic evaluation challenging.45
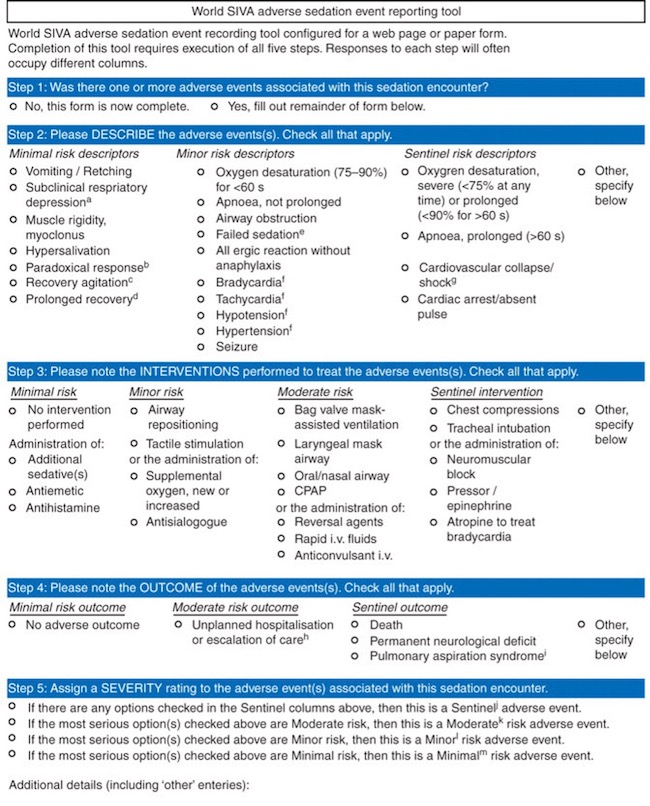
A simple, five-step adverse event reporting tool developed specifically for procedural sedation by the World Society of Intravenous Anesthesia’s (SIVA) International Sedation Task Force aims to simplify the process of medication error reporting (Figure 1). It includes standardized adverse event definitions which facilitates compiling and sharing data with researchers and quality stakeholders. Reporting systems like this can improve sedation practices and patient outcomes.45,46 The reporting tool can be accessed and submitted electronically at www.AESedationReporting.com. The five steps briefly described are as follows:
- Identify if adverse events were associated with sedation encounter.
- Characterize adverse events as minimal, minor, or sentinel risk.
- Characterize interventions performed to treat adverse events characterized as minimal, minor, moderate, or sentinel.
- Characterize the outcome adverse events as minimal, moderate or sentinel outcome.
- Assign a severity rating to the sedation encounter based on the most serious selection in prior steps.
Figure 1. World SIVA adverse sedation event-reporting tool.45
Data collected by event reporting systems are critical to improving medication safety in future instances of procedural sedation. Case-specific learning examples provide valuable feedback and a substrate for improvement so that future near misses or errors can be avoided.
Take Home Points
- Procedural sedation is a safe and effective method to facilitate painful and anxiety-provoking medical tests and procedures when administered by an experienced and trained provider and team.
- Pediatric patients undergoing procedural sedation pose unique challenges, most importantly weight-based dosing.
- Sedation providers must be able to readily identify common adverse side effects, perform emergency airway maneuvers, and administer reversal agents.
- Medication selection for procedural sedation should be guided by each clinical situation incorporating patient- and procedure-specific factors.
- Pre-procedure checklists and continuous monitoring should be routinely utilized during procedural sedation. The use of continuous waveform capnography to detect respiratory depression and apnea should be considered standard practice.
- Closed loop communication, use of common terminology and units, standardized dosing, syringe labeling, and adverse event reporting are evidence-based methods that can reduce the incidence of medication-related errors during procedural sedation.
Michael Leonardo Amashta, MD
PGY3 Resident Physician
Department of Emergency Medicine
UC Davis Health
mamashta@ucdavis.edu
David K. Barnes, MD, FACEP
Health Sciences Clinical Professor
Medical Co-Director
Department of Emergency Medicine
UC Davis Health
dbarnes@ucdavis.edu
References
- Green SM, Krauss B. Who owns deep sedation? Ann Emerg Med 2011;57(5):470–474. [Available at]
- Coté CJ, Wilson S. Guidelines for monitoring and management of pediatric patients during and after sedation for diagnostic and therapeutic procedures: an update.Pediatrics. 2006;118(6):2587–2602. [Free full text]
- Cravero JP, Blike GT, Beach M, et al. Incidence and nature of adverse events during pediatric sedation/anesthesia for procedures outside the operating room: report from the Pediatric Sedation Research Consortium.Pediatrics. 2006;118(3):1087-1096. [Available at]
- Green SM, Roback MG, Krauss BS, et al. Unscheduled procedural sedation: a multidisciplinary consensus practice guideline. Ann Emerg Med 2019;73(5):e51–e65. [Available at]
- Krauss B, Green SM. Procedural sedation and analgesia in children. Lancet. 2006;367(9512):766–780. [Available at]
- Gooden CK, Lowrie LH, Jackson BF, editors. The Pediatric Procedural Sedation Handbook. New York, NY: Oxford University Press; 2018. [Available at]
- American Society of Anesthesiologists [Internet]. Accessed February 15, 2023. Available at: https://www.asahq.org
- Coté CJ, Wilson S, American Academy of Pediatrics, American Academy of Pediatric Dentistry. Guidelines for Monitoring and Management of Pediatric Patients Before, During, and After Sedation for Diagnostic and Therapeutic Procedures. Pediatrics. 2019;143(6):e20191000. [Free full text]
- Sacchetti A, Senula G, Strickland J, et al. Procedural sedation in the community emergency department: initial results of the ProSCED registry.Acad Emerg Med. 2007;14(1):41-44. [Free full text]
- Kern J, Guinn A, Mehta P. Procedural sedation and analgesia in the emergency department.Emerg Med Pract. 2022;24(6):1–24. [Available at]
- Golparvar M, Saghaei M, Sajedi P, et al. Paradoxical reaction following intravenous midazolam premedication in pediatric patients – a randomized placebo-controlled trial of ketamine for rapid tranquilization. Pediatr Anesth. 2004;14(11):924–930. [Available at]
- Jackson BF, Beck LA, Losek JD. Successful flumazenil reversal of paradoxical reaction to midazolam in a child.J Emerg Med. 2015;48(3):e67–72. [Available at]
- Schofield S, Schutz J, Babl FE, Paediatric Research in Emergency Departments International Collaborative (PREDICT). Paediatric fracture reduction. Emerg Med Australas 2013;25(3):241–247. [Free full text]
- Elikashvili I, Vella AE. An evidence-based approach to pediatric procedural sedation. Pediatr Emerg Med Pract. 2012;9(8):1–16.
- Jalili M, Bahreini M, Doosti-Irani A, et al. Ketamine-propofol combination (ketofol) vs propofol for procedural sedation and analgesia: systematic review and meta-analysis. Am J Emerg Med. 2016;34(3):558–569. [Available at]
- Bhatt M, Johnson DW, Chan J, et al. Risk factors for adverse events in emergency department procedural sedation for children. JAMA Pediatr. 2017;171(10):957. [Free full text]
- Grunwell JR, Travers C, McCracken CE, et al. Procedural sedation outside of the operating room using ketamine in 22,645 children. Pediatr Crit Care Med. 2016;17(12):1109–1116. [Free full text]
- Josephy CP, Vinson DR. Feasibility of single- vs two-physician procedural sedation in a small community emergency department. Am J Emerg Med. 2018;36(6):977–982. [Available at]
- Vinson DR, Hoehn C. Sedation-assisted orthopedic reduction in emergency medicine: the safety and success of a one physician/one nurse model. West J Emerg. 2013;14(1):47–54. [Free full text]
- Clay-Williams R, Colligan L. Back to basics: checklists in aviation and healthcare. BMJ Qual Saf. 2015;24(7):428. [Free full text]
- Treadwell JR, Lucas S, Tsou AY. Surgical checklists: a systematic review of impacts and implementation. BMJ Qual Saf. 2014;23(4):299. [Free full text]
- Kahlenberg L, Harsey L, Patterson M, et al. Implementation of a modified WHO pediatric procedural sedation safety checklist and its impact on risk reduction. Hosp Pediatr. 2017;7(4):225–231. [Available at]
- Librov S, Shavit I. Serious adverse events in pediatric procedural sedation before and after the implementation of a pre-sedation checklist. J Pain Res. 2020;13:1797-1802. [Available at]
- Krauss B, Hess DR. Capnography for procedural sedation and analgesia in the emergency department. Ann Emerg Med. 2007;50(2):172-181. [Available at]
- Kron T. Stepping beyond the 5 Rights of Administering Drugs. Am J Nurs. 1962;62(7):62–63.
- Elliott M, Liu Y. The nine rights of medication administration: an overview. Br J Nurs. 2010;19(5):300–305. [Available at]
- Preventing Pediatric Medication Errors. The Joint Commission; 2021. Accessed April 4, 2023. [Free full text]
- Ghaleb MA, Barber N, Franklin BD, et al. Systematic review of medication errors in pediatric patients.Ann Pharmacother.2006;40(10):1766–1776. [Free full text]
- Hanna GM, Levine WC. Medication safety in the perioperative setting. Anesthesiol Clin. 2011;29(1):135–144. [Available at]
- Lenz JR, Degnan DD, Hertig JB, et al. A review of best practices for intravenous push medication administration. J Infusion Nurs. 2017;40(6):354–358. [Available at]
- Institute for Safe Medication Practices (ISMP).ISMP Safe Practice Guidelines for Adult IV Push Medications. ISMP;2015. Accessed April 4, 2023. [Free full text]
- Adapa RM, Mani V, Murray LJ, et al. Errors during the preparation of drug infusions: a randomized controlled trial. Br J Anaesth. 2012;109(5):729–734. [Free full text]
- Moreira ME, Hernandez C, Stevens AD, et al. Color-coded prefilled medication syringes decrease time to delivery and dosing error in simulated emergency department pediatric resuscitations. Ann Emerg Med. 2015;66(2):97-106.e3. [Free full text]
- Neuspiel DR, Taylor MM. Reducing the risk of harm from medication errors in children. Heal Serv Insights. 2013;6:HSI.S10454. [Free full text]
- Abdellatif A, Bagian JP, Barajas ER, et al. Look-Alike, Sound-Alike Medication Names: Patient Safety Solutions, Volume 1, Solution 1, May 2007. Jt Comm J Qual Patient Saf. 2007;33(7):430–433. [Available at]
- Institute for Safe Medication Practices (ISMP).ISMP List of Confused Drug Names. ISMP; 2019. Accessed February 15, 202. [Free full text]
- Do Not Use List Fact Sheet [Internet]. The Joint Commission. Accessed Feb 15, 2023. [Free full text]
- Ostini R, Roughead EE, Kirkpatrick CMJ, et al. Quality Use of Medicines – medication safety issues in naming; look‐alike, sound‐alike medicine names. Int J Pharm Pract. 2012;20(6):349–357. [Available at]
- Brown JP. Closing the communication loop: using readback/hearback to support patient safety.Jt Comm J Qual Saf. 2004;30(8):460–464. [Available at]
- Karamnov S, Sarkisian N, Grammer R, et al. Analysis of adverse events associated with adult moderate procedural sedation outside the operating room. J Patient Saf. 2017;13(3):111–121. [Available at]
- Kilner E, Sheppard LA. The role of teamwork and communication in the emergency department: a systematic review. Int Emerg Nurs. 2009;18(3):127–137. [Available at]
- Doorey AJ, Turi ZG, Lazzara EH, et al. Safety gaps in medical team communication: Results of quality improvement efforts in a cardiac catheterization laboratory. Cathet Cardiovasc Intervent. 2020;95(1):136–144. [Available at]
- Rishoej RM, Hallas J, Kjeldsen LJ, et al. Likelihood of reporting medication errors in hospitalized children: a survey of nurses and physicians. Ther Adv Drug Saf. 2017;9(3):179–192. [Free full text]
- Sanghera IS, Franklin BD, Dhillon S. The attitudes and beliefs of healthcare professionals on the causes and reporting of medication errors in a UK intensive care unit. Anaesthesia. 2007;62(1):53–61. [Free full text]
- Mason KP, Green SM, Piacevoli Q; International Sedation Task Force. Adverse event reporting tool to standardize the reporting and tracking of adverse events during procedural sedation: a consensus document from the World SIVA International Sedation Task Force. Brit J Anaesth. 2012;108(1):13–20. [Free full text]
- Bhatt M, Kennedy RM, Osmond MH, et al. Consensus-based recommendations for standardizing terminology and reporting adverse events for emergency department procedural sedation and analgesia in children.Ann Emerg Med. 2009;53(4):426-435.e4. [Available at]