One Bronchoscopy, Two Errors
Leiten E, Nielsen R. One Bronchoscopy, Two Errors. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2019.
Leiten E, Nielsen R. One Bronchoscopy, Two Errors. PSNet [internet]. Rockville (MD): Agency for Healthcare Research and Quality, US Department of Health and Human Services. 2019.
The Case
A 67-year-old man with a history of hypertension was admitted to the intensive care unit (ICU) with hypoxic respiratory failure secondary to community-acquired pneumonia. For his severe hypoxia, he was managed with high-flow nasal cannula and did not require mechanical ventilation. He was given intravenous fluids and antibiotics.
On the second hospital day, the patient had increased pulmonary secretions. The critical care provider decided to perform bronchoscopy at the bedside. After informed consent and a procedural time out, the patient was given 2 mg of intravenous midazolam, a sedating agent. Once he was lightly sedated, a flexible bronchoscope was introduced.
The airways were inspected throughout. Then, per usual protocol, bronchoalveolar lavage was performed by introducing 20 mL of normal saline to the right middle lobe. This fluid was suctioned out and sent to the laboratory for analysis.
The bronchoscope was removed without any complications. However, the patient was difficult to arouse after the procedure. This was unexpected given that only a small amount of midazolam had been used. The patient was nearly apneic with very shallow breaths and the decision was made to intubate. The patient did not need any further sedation during the intubation.
The entire care team paused in real time to review the events. The critical care provider noticed that the syringes that contained the different fluids were not labeled and were both stored on the same shelf of the procedure cart. It was discovered that after the 2 mg of intravenous midazolam was given, instead of flushing this with normal saline, it had been flushed with an additional 10 mg of midazolam (the nurse had flushed with 2 mL and the midazolam concentration was 5 mg/mL). This high dose of midazolam had led to the respiratory failure requiring intubation.
On top of that, instead of normal saline, lidocaine had been used for the lung lavage. The lidocaine was available in a syringe for the bronchoscopy, as occasionally patients have severe discomfort or coughing from the bronchoscope and the lidocaine can be used to treat this. The lidocaine syringe was also not labeled and was sitting next to the syringe with normal saline. Fortunately, the patient did not experience any adverse consequences from the use of the lidocaine for the lavage (high doses of lidocaine can enter the systemic circulation and lead to neurological and cardiovascular adverse effects).
The patient remained intubated until the midazolam wore off. He woke up and was able to be extubated safely later that day. He slowly improved with treatment of his pneumonia and went home a few days later. The care team disclosed the errors to the patient including a commitment to making improvements to prevent such an error in the future.
The Commentary
by Elise Orvedal Leiten, MD, and Rune Nielsen, MD, PhD
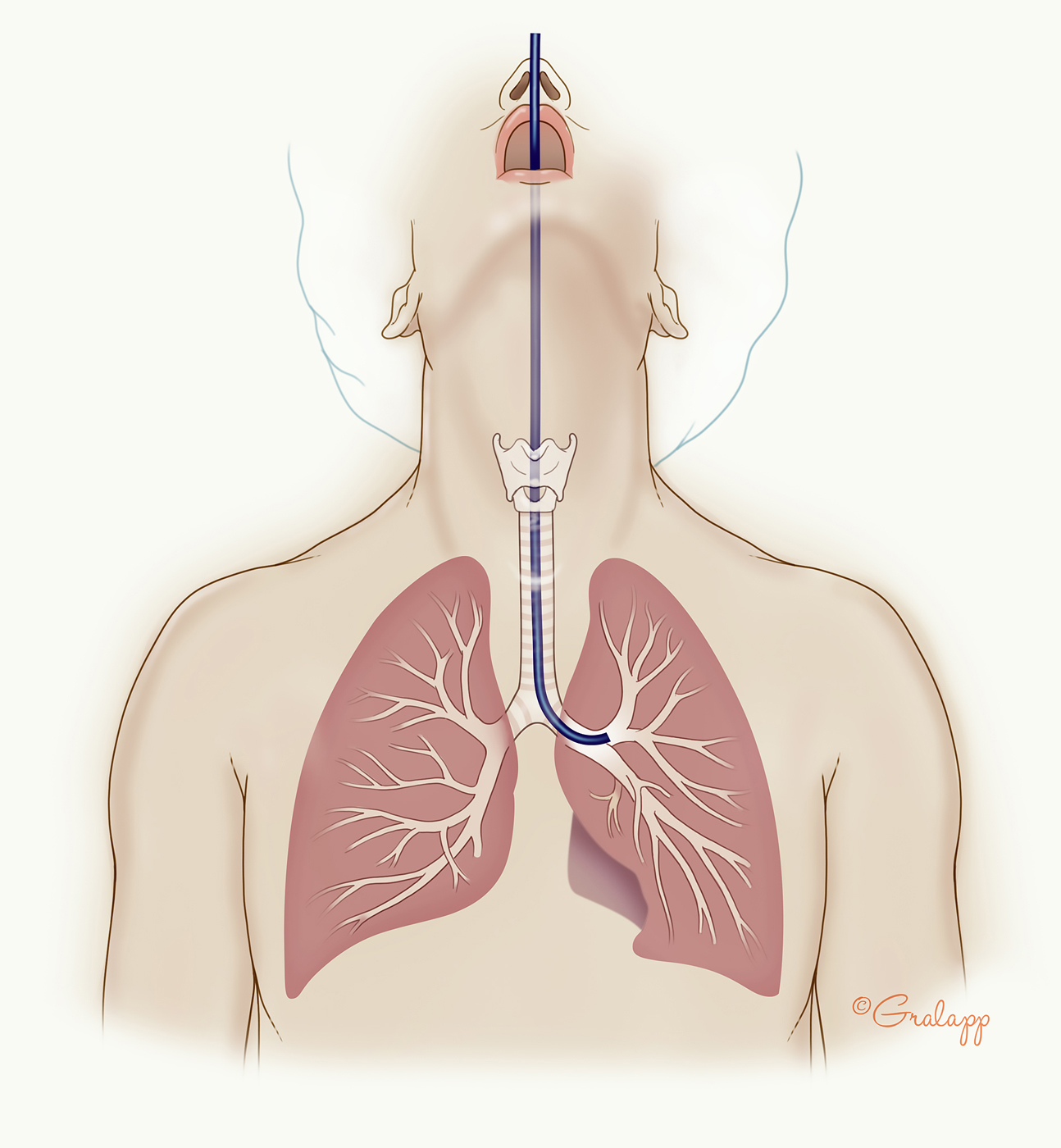
Bronchoscopy (Figure 1) is performed for a wide range of diagnostic, therapeutic, and research purposes, and it is regarded as a cornerstone of pulmonary medicine.(1) Indications for bronchoscopy include (but are not restricted to) diagnosis of respiratory infections, diagnosis of lung or mediastinal tumors/masses/infiltrates, evaluation of interstitial lung disease, and a large variety of therapeutic procedures such as foreign body removal, relief of mucus plugging, stenting procedures, bronchoscopic lung volume reduction, endobronchial tumor debulking (laser, brachytherapy), and bronchial thermoplasty for severe asthma.
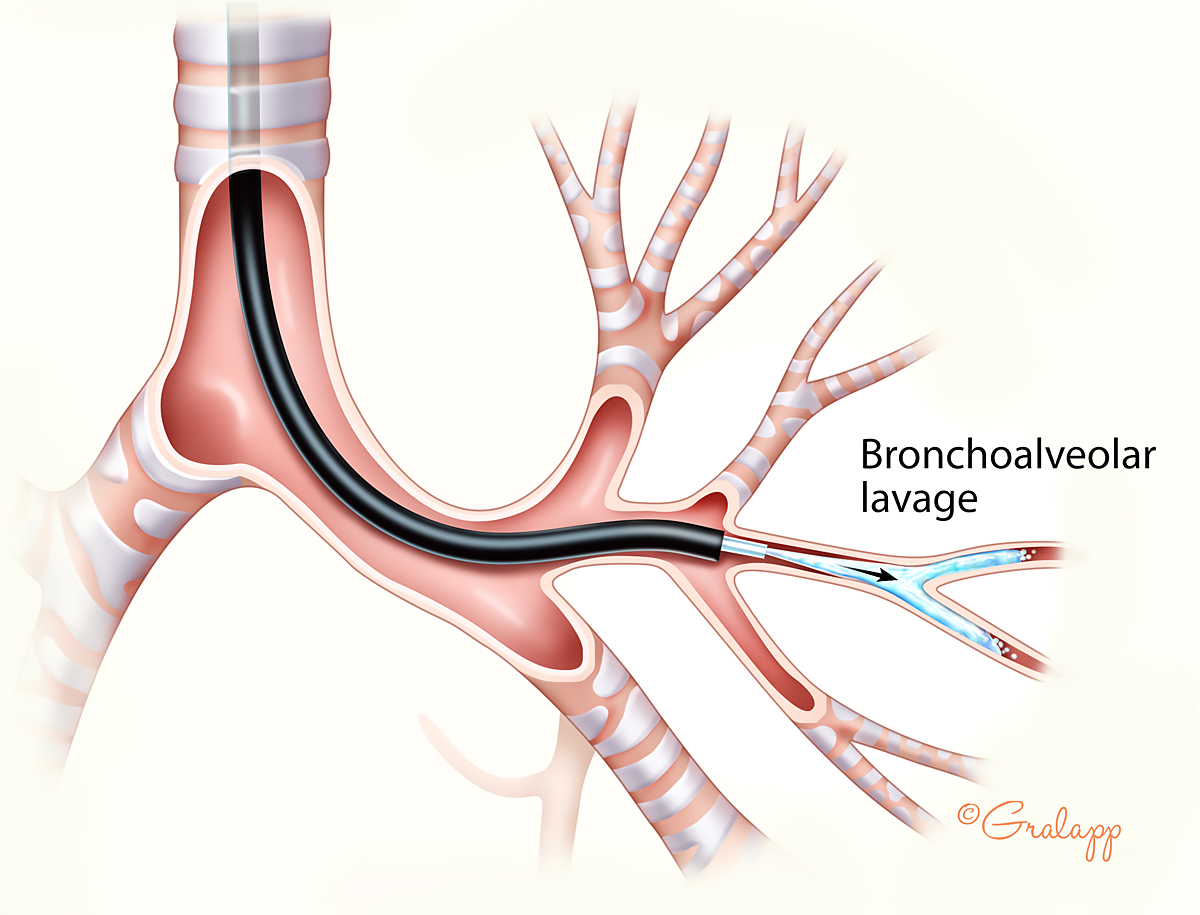
Bronchoscopy can be performed in a number of ways, with either a flexible or rigid scope, and can be done at the bedside or in the bronchoscopy suite. Several types of visualization, sampling, and treatment techniques can be applied, and the procedure can be performed with no, light, moderate, or deep sedation/general anesthesia.(1) In this case, flexible bronchoscopy was part of a diagnostic workup for pneumonia. The procedure included small-volume lavage (SVL), where 20 mL saline was instilled to the airways via the working channel of the bronchoscope, and then retrieved and investigated. Full bronchoalveolar lavage (BAL) (Figure 2) usually involves larger volumes (typically > 100 mL). We do not wish to evaluate the choice of lavage in this case, since no current recommendation exists and we have limited information regarding the patient. In general, SVL can be seen as favorable to BAL in the case of severe lung disease, as the load of fluid installed is smaller. In cases of nonresolving pneumonia, BAL and SVL can assist in the search of an etiologic diagnosis (2), particularly in immune-compromised patients (3) where detection of Pneumocystis jiroveci, Mycobacterium tuberculosis, and Aspergillus species is crucial for the choice of antimicrobial therapy. The intent was to place the patient in this case under light sedation with midazolam. In most centers, light sedation can be performed without the supervision of an anesthesiologist or nurse anesthetist.(3)
Common complications of diagnostic bronchoscopy relate to the bronchoscope's irritation of the respiratory tract (cough, bronchospasm), sampling procedures (bleeding, pneumothorax), and sedation (desaturation, hypotension, loss of consciousness). Severe complications requiring intervention or resulting in increased mortality or morbidity are rare. While the procedure is generally safe, it is often associated with discomfort, and many patients experience fever following bronchoscopy.(4)
In an effort to improve bronchoscopy safety, the British Thoracic Society (BTS) has developed a guideline for diagnostic flexible bronchoscopy in adults. The guideline recommends bronchoscopist training and the use of checklists.(3) A consensus statement (5) on bronchoscopy training suggests that professional societies move from volume-based certification systems (e.g., certification happens after the completion of x procedures) to skill acquisition and knowledge-based competency assessment. They also recommend that bronchoscopy training programs should include simulation. Further, they recommended introduction of quality and process improvement systems and regular assessment of skill maintenance after skill acquisition.
The British Thoracic Society's recommended bronchoscopy safety checklist is adapted from the World Health Organization (WHO) surgical safety checklist (6), and it is divided into three checkpoint sections: before bronchoscopy, before sedation, and after the procedure. The checklist (Figure 3) is not intended to be comprehensive, and local modifications and additions are encouraged.(3) The error presented in the case could not have been avoided simply with adherence to the original checklist, as the only checkpoint regarding medication ("Have all instruments and controlled drugs been accounted for") is first mentioned in the "After the procedure" section. At our unit, all drugs are labeled before procedures using prelabeled adhesive stickers.
In this case, the bronchoscopy was performed in an ICU setting. The British Thoracic Society guideline for diagnostic flexible bronchoscopy in adults outlines a few key principles related to bronchoscopy in the ICU. The guideline states that "patients in the ICU should be considered at high risk from complications when undergoing bronchoscopy."(3) It gives the following recommendations for the ICU setting: "Continuous multimodal physiological monitoring should occur during and after bronchoscopy," "when patients require non-invasive ventilation prior to bronchoscopy, the procedure should be conducted in an environment where intubation and ventilatory support are readily accessible," and "clinicians administering sedation/anesthesia/analgesia should be acquainted with the use of these agents, and the anesthetist/intensivist is usually best placed to fulfil this role."(3) In this case, it is not clear if there was multimodal physiologic monitoring, but there was equipment available for intubation. It is assumed that the intensivist was familiar with midazolam, but it is not clear what training he had in sedation.
When reviewing the medical literature, we did not find any statistical overview nor any case reports directly concerning medical errors in bronchoscopy. There are no scientific publications on syringe labeling errors or sedation dose errors in bronchoscopy specifically, although there have been discussions of syringe labeling errors in other settings, including in this series.(7-9) These commentaries highlight immediate labeling of syringes, discarding of unlabeled syringes, routine double checks, and prefilled barcoded syringes as means to avoid IV drug administration errors. In other words, it seems likely that similar cases in bronchoscopy have occurred in the past but have not been reported.
The British Thoracic Society guideline for diagnostic flexible bronchoscopy does make recommendations that, if followed, could potentially have prevented this error. Specifically, the guideline recommends that only 1 mg/mL vials are available in bronchoscopy suites. Larger 2 mg/mL or 5 mg/mL vials should not be made available without a formal risk assessment.(3) In this case, bronchoscopy was conducted in the ICU and not in the bronchoscopy suite. Still, limiting the availability of high-dosage vials in the bronchoscopy environment could reduce the potential harm of this accident.
A side note: The most noticeable error in this case was the overdose of midazolam, as it led the patient into respiratory failure requiring intubation. The second error, using high dosages of lidocaine instead of saline, could have resulted in cardiovascular or neurological adverse events. It is possible that the second error would have gone unnoticed if it weren't for the first error. Although the patient did not experience any direct harm from the lidocaine, he could have experienced harm indirectly if the error remained undiscovered. Lidocaine has antimicrobial properties and has been shown to reduce the growth of certain bacterial species when added to BAL fluid.(10) Had the error in this case not been discovered, the BAL results could have led to a change of treatment not benefiting the patient.
In order to prevent errors in the bronchoscopy setting, we recommend that all institutions adhere to the British Thoracic Society guideline. As encouraged by the guideline, each institution should make modifications and additions to the WHO safety checklist to fit local practices. As of today, it is not clear which system checks should be put into place. We argue that errors and situations identified as prone to error should be reported to case review committees and evaluated. Documentation of errors can assist the development of more precise checklists and might also improve the training of bronchoscopists. Bronchoscopists should undergo simulation-based training, and certifications should be based on skills and knowledge instead of volume.
Take-Home Points
- Diagnostic bronchoscopy and bronchoalveolar lavage sampling is generally safe.
- Like any other invasive procedure, bronchoscopy is associated with potential complications. Therefore, high-risk patients (such as those in the ICU) should not undergo bronchoscopy without a clear indication and an assessment of the risks and benefits.
- Drug administration errors can occur in the setting of bronchoscopy, can be fatal, and are probably underreported. Adherence to guidelines and systematic approaches to improve drug administration procedures can reduce the amount and harm of errors. Documentation of errors might improve checklists and bronchoscopy training.
Elise Orvedal Leiten, MD
Department of Clinical Science
University of Bergen
Bergen, Norway
Rune Nielsen, MD, PhD
Associate Professor
Department of Clinical Science
University of Bergen
Bergen, Norway
References
1. Ernst A. Introduction to Bronchoscopy. 1st ed. Cambridge: Cambridge University Press; 2009. ISBN: 9780521766289.
2. van der Eerden MM, Vlaspolder F, de Graaff CS, Groot T, Jansen HM, Boersma WG. Value of intensive diagnostic microbiological investigation in low- and high-risk patients with community-acquired pneumonia. Eur J Clin Microbiol Infect Dis. 2005;24:241-249. [go to PubMed]
3. Du Rand IA, Blaikley J, Booton R, et al; British Thoracic Society Bronchoscopy Guideline Group. British Thoracic Society guideline for diagnostic flexible bronchoscopy in adults: accredited by NICE. Thorax. 2013;68(suppl 1):i1-i44. [go to PubMed]
4. Leiten EO, Martinsen EM, Bakke PS, Eagan TM, Grønseth R. Complications and discomfort of bronchoscopy: a systematic review. Eur Clin Respir J. 2016;3:33324. [go to PubMed]
5. Ernst A, Wahidi MM, Read CA, et al. Adult bronchoscopy training: current state and suggestions for the future: CHEST Expert Panel Report. Chest. 2015;148:321-332. [go to PubMed]
6. Mahajan RP. The WHO surgical checklist. Best Pract Res Clin Anaesthesiol. 2011;25:161-168. [go to PubMed]
7. Weingart SN. It's all in the syringe. AHRQ WebM&M [serial online]. August 2006. [Available at]
8. Barach P. Unexplained apnea under anesthesia [Spotlight]. AHRQ WebM&M [serial online]. February 2003. [Available at]
9. Kulli JC. Pocket syringe swap. AHRQ WebM&M [serial online]. May 2011. [Available at]
10. Olsen KM, Peddicord TE, Campbell GD, Rupp ME. Antimicrobial effects of lidocaine in bronchoalveolar lavage fluid. J Antimicrob Chemother. 2000;45:217-219. [go to PubMed]
Figures
Figure 1. Bronchoscopy. (Illustration © 2018 Chris Gralapp.)
Figure 2. Bronchoalveolar Lavage. (Illustration © 2018 Chris Gralapp.)
Figure 3. British Thoracic Society's Recommended Bronchoscopy Safety Checklist.(3) Click on thumbnail for PDF of Figure